Solid-state electrolytes, such as those made from ceramic-polymer composites, are promising alternatives to liquid electrolytes for next-generation lithium-ion batteries that are safer and more stable. However, there is a problem in that these composites agglomerate at high weight ratios, which adversely affects their good ion conductivity properties. A new 3D interconnected framework consisting of ceramic nanofillers made using a nanostructured hydrogel template that does not aggregate could help overcome these problems.
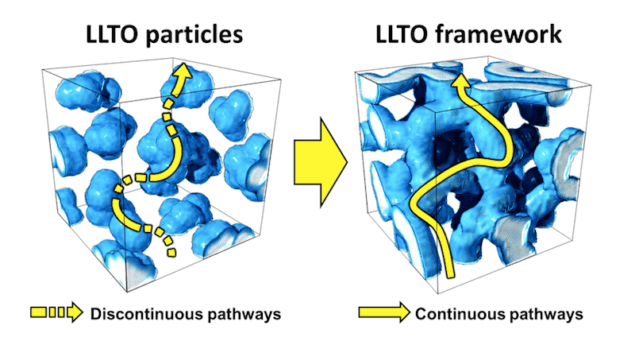
“Ceramic-polymer composite electrolytes comprising ceramic nanofillers and a polymer matrix have improved mechanical and thermal stability as well as ionic conductivities that are better than pure polymer electrolytes,” explains Guihua Yu of the University of Texas at Austin, who led this study. “However, as these composites agglomerate at high weight ratios, the ion conductivity pathways in these materials becomes discontinuous. This is because the percolating network in the nanofiller deteriorates.
“In this new work, we fabricated 3D interconnected frameworks consisting of ceramic nanofillers using a simple nanostructured-hydrogel template technique. The framework provides a continuous ion conduction path that prevents nanofiller aggregation. It also provides excellent conductivity and electrochemical stability.”
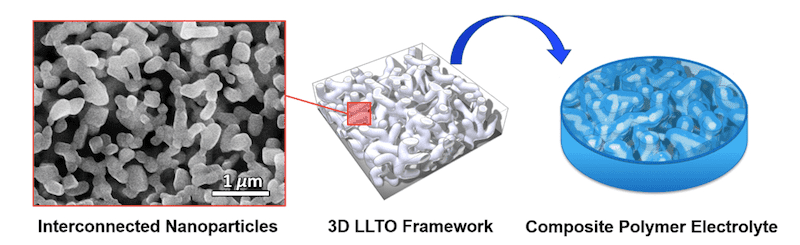
Yu and colleagues made their percolating network of ceramic nanoparticles by nanoscale phase separation of a polymer (polyethylene oxide), water, and ceramic precursors (Li0.35La0.55TiO3, or LLTO). The nanosized ceramic particles are interconnected to form a free-standing continuous, 3D porous structure.
“Once the self-supporting ceramic framework has formed, we fill the void pores with the polymer to prepare a polymer electrolyte,” Yu tells nanotechweb.org. “Unlike traditional composites in which particles are dispersed and separated in the polymer matrix, the nanostructured framework acts as a 3D nanofiller that can then form a continuous interphase between the polymer and the ceramic. The interaction between the filler and the polymer results in improved lithium ion conductivity, as mentioned, thanks to the formation of a continuous interphase, but also in sufficient mechanical integrity for use in a variety of flexible electronic devices.”
According to Yu, the interphase between the ceramic filler and the polymer is responsible for faster lithium ion conduction thanks to the high dielectric constant and the abundant surface defects in the LLTO.
The team, reporting its work in Angewandte Chemie DOI: 10.1002/anie.201710841, says that it would now like to apply its structural design strategy to other energy applications, such as composite electrodes. To this end, we need to better understand the interaction between the ceramic filler and the polymer matrix,” explains Yu.



