A team from the UK and Spain has found that pH and the prevalence of organic matter in soil affect the binding of cobalt with minerals and organo-mineral complexes. This has implications for the clean-up of contaminated land.
Using synchrotron x-ray absorption spectroscopy, Gemma Woodward and colleagues identified a universal uptake mechanism for cobalt in soil and sediment.
The team showed that where the soil contains lots of organic matter, as is the case for peat, cobalt binds more easily to the surface of the soil complex. Although binding to the soil makes cobalt more likely to affect humans near a spill, it improves prospects for remediation that collects and treats soil.
If there is little organic matter in the soil, however, the cobalt remains mobile and is more likely to enter groundwater and rivers. Cobalt is highly soluble so a cobalt pollution event of this type could be challenging to mitigate.
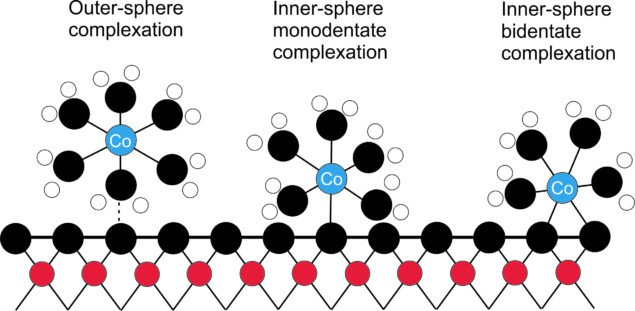
The researchers found that cobalt behaviour also varies with pH. At low pH cobalt forms a relatively weak electrostatic bond with the soil known as outer-sphere complexation. Whereas at high pH an inner-sphere bidentate complex forms via two stronger covalent bonds. For minerals containing iron, there was no bonding at low pH. That indicates that in iron-rich soils, cobalt contamination would remain mobile and potentially spread over a larger area.
“These findings mean that at pH 6.5 or lower cobalt will be mobile in deserts and alluvial sediments that contain little organic matter, whereas on peat, cobalt will be mobile only at very low pH,” says Woodward.
In natural environments a combination of organic matter and minerals is most likely and cobalt binding will probably be an amalgam of both conditions. “Artificial remediation can be very expensive, so where cobalt is mobile, supporting natural attenuation processes may be more appropriate and effective,” Woodward adds.
Cobalt is a heavy metal that enters the environment from metal production facilities, coal power plants or vehicle emissions. A radioactive form, cobalt-60, is a by-product of nuclear reactions and is found in the cooling waters of nuclear power plants. In small amounts, cobalt is beneficial to plants but high concentrations stop plants producing enough chlorophyll. The gamma radiation emitted by cobalt-60 is useful in radiotherapy and radiography, but cobalt may cause heart disease in humans.
Woodward and colleagues published their study in Geochimica et Cosmochimica Acta.