Damage to the spinal cord, whether by injury or disease, can have devastating impacts on health, including loss of motor or sensory functions, or chronic back pain, which affects an estimated 540 million people at any given time. A US-based research team has now used functional ultrasound imaging (fUSI) to visualize the spinal cord and map its response to electrical stimulation in real time, an approach that could improve treatments of chronic back pain.
Despite playing a central role in sensory, motor and autonomic functions, little is known about the functional architecture of the human spinal cord. Traditional neuroimaging techniques, such as functional MRI (fMRI), are impeded by strong motion artefacts generated by heart pulsation and breathing.
In contrast, fUSI is less impacted by motion artefacts and can image the spinal cord with high spatiotemporal resolution (roughly 100 µm and up to 100 ms) and high sensitivity to slow flowing blood during surgery. It works by emitting ultrasonic waves into an area-of-interest and detecting the echoed signal from blood cells flowing in that region (the power Doppler signal). Another advantage is that the fUSI scanner is mobile, eliminating the extensive infrastructure required for fMRI systems.
“The spinal cord houses the neural circuitry that controls and modulates some of the most important functions of life, such as breathing, swallowing and micturition. However, it has been frequently neglected in the study of neural function,” explains lead contact Vasileios Christopoulos from the University of California Riverside. “Functional ultrasound imaging overcomes the limitations of traditional neuroimaging technologies and can monitor the activity of the spinal cord with higher spatiotemporal resolution and sensitivity than fMRI.”
Previous research demonstrated that fUSI can measure brain activity in animals and human patients, including one study showing that low-frequency fluctuations in the power Doppler signal are strongly correlated with neuronal activity. More recently, researchers used fUSI to image spinal cord responses to electrical stimulation in animals.
In this latest work, Christopoulos and colleagues – also from the USC Neurorestoration Center at the Keck School of Medicine – used fUSI to characterize haemodynamic activity (changes in blood flow) in the spinal cord in response to epidural electrical spinal cord stimulation (ESCS) – a neuromodulation tool employed to treat pain conditions that don’t respond to traditional therapies.
In a first in-human study, the team monitored haemodynamic activity in six patients undergoing implantation of a therapeutic ESCS device to treat chronic back pain, reporting the findings in Neuron.
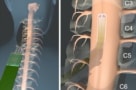
Utilizing a similar mechanism to fMRI, fUSI relies on the neurovascular coupling phenomenon, in which increased neural activity causes localized changes in blood flow to meet the metabolic demands of active neurons. The team used a miniaturized 15-MHz linear array transducer to perform fUSI, inserting it surgically onto the spinal cord at the tenth thoracic vertebra (T10), with the stimulation electrodes placed to span the T8–9 spinal segments. The recorded images had a 100 x 100 µm spatial resolution, a slice thickness of about 400 µm and a 12.8 x 10 mm field-of-view.
Four patients received 10 ON–OFF cycles of low-current (3.0 mA) stimulation, comprising 30 s with stimulation then 30 s without. Stimulation caused regional changes in spinal cord haemodynamics, with some regions exhibiting significant increases in blood flow and others showing significant decreases. Once the stimulation was switched off, blood flow returned to the initial condition.
To assess whether fUSI can detect haemodynamic changes associated with different stimulation protocols, the remaining two patients received five ON–OFF cycles of 3.0 mA stimulation followed by five cycles of 4.5 mA stimulation, with a 3-min pause between the two. The researchers found that increasing the current amplitude from 3.0 to 4.5 mA did not change the spatial distribution of the activated spinal cord regions. However, high-current stimulation induced stronger haemodynamic changes on the spinal cord.
This ability of fUSI to differentiate haemodynamic responses evoked by different ESCS currents is an important step towards developing an ultrasound-based clinical monitoring system to optimize stimulation parameters. Christopoulos explains that because patients are anaesthetized during spinal cord surgery, they cannot report whether the applied electrical stimulation protocol actually reduces pain. As such, the neurosurgeon cannot accurately assess the effects of neuromodulation in real-time.
Microelectrode array could enable safer spinal cord surgery
“Our study provides a first proof-of-concept that fUSI technology can be used to develop closed-loop clinical neuromodulation systems, allowing neurosurgeons to adjust stimulation parameters (pulse width, pulse shape, frequency, current amplitude, location of stimulation, etc) during surgery,” he tells Physics World.
In future, the team hopes to establish fUSI as a platform for investigating spinal cord function and developing real-time closed-loop clinical neuromodulation systems. “We recently submitted for publication a clinical study demonstrating that fUSI is capable of detecting networks in the human spinal cord where activity is strongly correlated with bladder pressure,” says Christopoulos. “This finding opens new avenues for the development of spinal cord machine interface technologies to restore bladder control in patients with urinary incontinence, such as those with spinal cord injury.”