
Drugs affect the metabolism of cells by inhibiting the activity of specific proteins. To do so, a drug binds to a target protein, in a pouch-like functional compartment of the protein. This binding process can also alter the structure of the protein surface, a phenomenon called conformational change, and open new pouches that would be good nesting spots for active agents.
Taking advantages of these new pouches is a holy grail of drug research, as they are likely to offer better selectivity of active agents and reduce side effects. However, conventional biophysical methods for monitoring the protein surface are currently too limited to enable such breakthroughs. While X-ray crystallography and NMR spectroscopy are highly efficient, they are also time and material consuming and need weeks to yield results. A new tool for quick monitoring of the structure of the protein is hence needed.
Tracking conformational changes
Klaus Gerwert and his team at Ruhr-Universität Bochum addressed this challenge by using an infrared (IR) sensor able to track protein conformational changes in just a few minutes. The sensor is based on a crystal that is permeable for IR light and allows tracking of the absorbance spectra.
When a protein binds on the surface of the sensor, it is then washed by a liquid containing the active agent and the recorded spectrum is analysed. The correlation between the IR spectrum and the protein structure enables direct molecular interpretation of the spectrum and study of conformational changes induced by the active agent (Ang. Chem. Int. Ed. 57 9955).
To investigate the sensor’s application, the researchers inhibited the heat shock protein 90 (HSP90), which assists newly translated proteins in the cell to fold in the correct 3D structure and is required by tumour cells to grow rapidly. They particularly looked at the changes induced by different compounds in HSP90 that are known to use various binding modes to drugs.
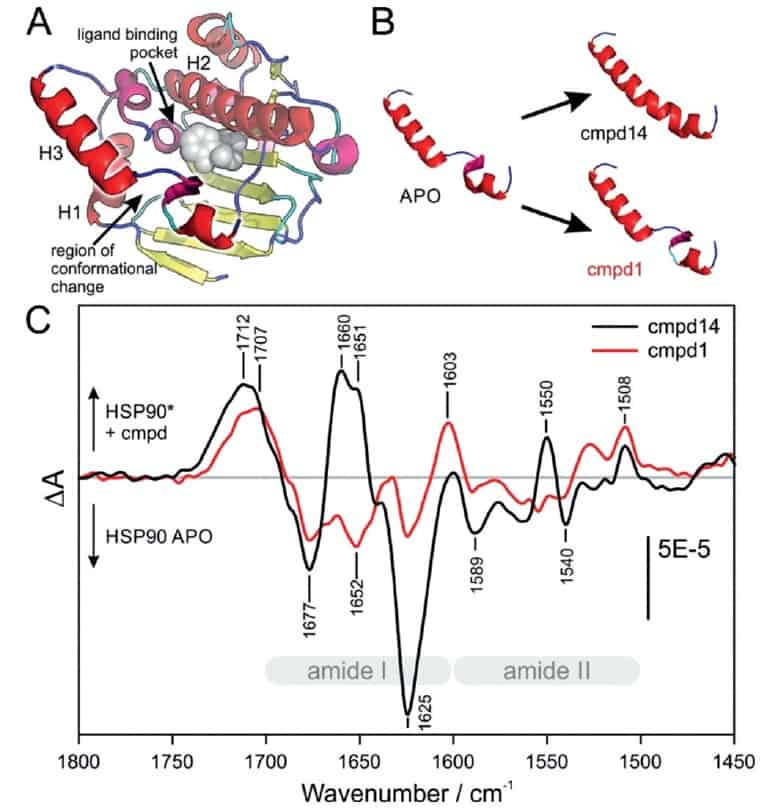
The conclusions drawn from the absorbance spectra matched those obtained from X-ray data and clearly showed that the sensor could be used to track conformational changes. The researchers further validated the technique by measuring the absorbance of several compounds interacting together at the same time and identifying their respective binding modes. They then compared the results against both theory and subsequent crystallography experiments. In both cases, the sensor was able to identify the correct binding mode, even when the conformational differences were small.
Manufacturing more efficient drugs
The efficacy period of a drug in the body corresponds to the rate at which its molecules disengaged from the target protein. The longer an active agent is bound to the target protein, the more effective it is and the less side effects it causes. By providing near-real-time information about the interaction between a drug and its target protein, the sensor-based technique could help pharmaceutical companies to design new and more efficient drugs.



