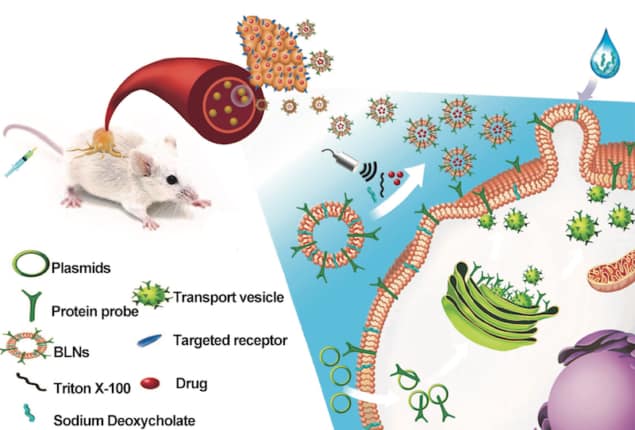
Although precision medicine is coming along in leaps and bounds, targeted drug delivery to tumours remains a huge challenge. A variety of nanomaterials, including ligand-conjugated liposomes, exosomes, inorganic nanoparticles and immunoliposomes, can be used to deliver specific drugs but the problem is that it is difficult to maintain the bioactivity of these therapeutics. What is more, they must be oriented in the correct way to efficiently target a cell population of interest. A team of researchers at Xiamen University in China has now developed a biomimetic theranostic ligand-targeted nanoplatform based on bio-functionalized liposome-like nanovesicles (BLNs) containing protein/peptide ligands that overcomes all these problems.
Personalized, or precision, medicine will replace the traditional and outdated “one size fits all” medical approach to one that will be safe, precise and tailored to each individual. Precision oncology or precision medicine of cancer aims to match the most accurate and effective treatment to each patient.
Researchers led by Gang Liu at the Innovation Center for Molecular Imaging and Translational Medicine at Xiamen University say they developed their BLNs to selectively target a wide spectrum of specific cell populations as well as tumour tissue thanks to appropriately-designed protein probes on their surface. “These ligands can be affibodies, single-chain antibodies or nanobodies,” explains Liu. “In our study, and as a proof of concept, we genetically engineered nanovesicles displaying human epidermal growth factor (hEGF) or affibody ligands that target tumour cells.”
Ligands maintain their good bioactivity
The researchers began by engineering natural hGEF or affibody ligands so that they could be guided to the cellular surface. They then triggered a process that caused giant plasma membrane vesicles to bud from the cell surface in a way that is very similar to how exosomes – tiny membrane-bound spheres – are excreted from cells. These vesicles help in the formation of nanoscale BLNs functionalized with protein ligands that are oriented in the right direction to better target tumour cells, says Liu.
“And that is not all: we inserted the protein probes into vesicular membranes using intracellular biosynthetic routes rather than by chemical conjugation techniques,” he adds. “This allows the ligands to maintain their good bioactivity because it avoids vulnerable biomacromolecules from being lost through non-specific covalent linking processes.”
Better than liposomal doxorubicin
The team tested nanovesicles containing affibody ligands on two types of tumour cells and BT474 tumour xenograft models, and found that those loaded with the widely-used anti-cancer drug doxorubicin were much better anti-cancer agents than the clinically approved liposomal doxorubicin.
“Doxorubicin-loaded nanovesicles displaying affibody targeting moieties can be more effectively bound and taken up by HER2-overexpressing tumour cells via a mechanism called specific receptor-mediated endocytosis,” explains Liu. “As a result, enhanced Dox accumulation in HER2-overexpressing tumours substantially improves the antitumour efficacy resulting from BLN targeting delivery, and importantly, also significantly reduces the Dox distribution in major vital organs, which is a problem for many antitumour therapies.”
Even though BLNs might be limited when compared to some synthetic nanocarriers, such as block copolymer micelles (because they may be difficult to purify or reproduce), they could still make good nanoplatforms for producing a wide range of ligand-targeted nanovesicles, he adds.
“They could also be adapted to design analogue vehicles for drug-targeted delivery to a wide range of specific cell populations and tissues by engineering the appropriate protein probes on the BLN surface.”
The nanovesicles are described in Advanced Materials DOI: 10.1002/adma.201705350.