Microwave breast imaging (MBI) represents a promising non-invasive technology for detection of breast tumours. It does not utilize ionizing radiation nor require breast compression, potentially offering a safer and more comfortable method for breast cancer screening.
As of 2020, 10 MBI prototypes had been clinically tested. Researchers at Galway University Hospital and the National University of Ireland Galway have now performed first-in-human testing of an eleventh: the Wavelia system developed by MVG Industries. The clinical investigation, described in Academic Radiology, demonstrated Wavelia’s ability to detect and localize breast tumours, and to quantify the accuracy of lesion localization. The system proved to be safe and was well tolerated by the 25 women participating in this evaluation.
MBI is based on the differences in the electrical properties of malignant and normal breast tissue at microwave frequencies. Cancerous tissue has increased water content and vascularity compared with healthy tissue, resulting in a higher dielectric constant. MBI can quantify the location, shape and/or morphological properties of lesions based on this dielectric contrast. The measured data are then inverted by an imaging algorithm to produce an image of the breast.

The Wavelia prototype includes two subsystems, the optical breast contour detection (OBCD) subsystem and the MBI subsystem, each integrated in an identical examination table. The OBCD subsystem consists of a 3D stereoscopic camera placed below the table, which is scanned to reconstruct the external surface of the breast and calculate breast volume, with the patient lying in prone position. The MBI scan is then performed using low-power, non-ionizing microwaves that propagate through the breast.
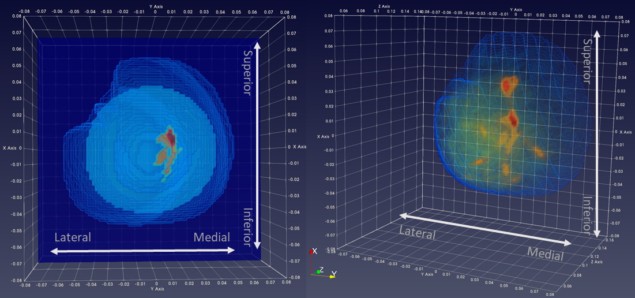
The Wavelia MBI system uses 18 equally-spaced wideband Vivaldi-type antennas arranged in a horizontal circle outside a cylinder containing coupling fluid. Each probe illuminates the imaging domain in turn, while the remaining antennas receive the electromagnetic scattering at various angles around the circle. The probe array also moves vertically, illuminating the breast at 5 mm intervals to capture the dielectric contrast for the entire breast. The system uses the MBI data to generate 10 mm-thick coronal sections of the breast, and then integrates the partially overlapping consecutive sections to form a 3D MBI image.
Clinical investigation
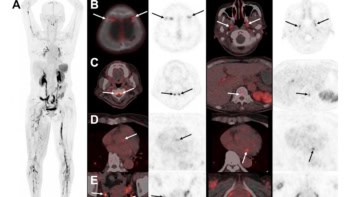
Lead author Brian Moloney and co-authors analysed MBI scans acquired from 25 patients, with 24 included in the final data analysis. The cohort included 11 patients with invasive carcinoma (six cases of invasive ductal carcinoma (IDC) and five of invasive lobular carcinoma (ILC)), plus four cases of benign breast disease, eight patients with a breast cyst and one with a complicated cyst.
MBI detected four of the six IDC lesions and all of the ILC lesions, correctly localizing seven of the nine detected cancerous lesions. The two IDCs that were not detected were less than 10 mm in size. Of the 13 benign cases, MBI detected 12 lesions and accurately approximated the location for 10 of these.
The patients also participated in a survey assessing the acceptability of the MBI process. This included questions regarding the scanning time, system vibration and noise, the temperature and smell of the surrounding liquid, and the comfort of the examination table. They were also asked whether they would recommend the procedure to other women. Responses were favourable, with 92% reporting that they would recommend the procedure.
Importantly, no clinical trial-related adverse events were recorded, highlighting the safety profile of the Wavelia MBI system.
Based on feedback from this study, the team developed a second Wavelia prototype. Technical upgrades include: an enlarged opening in the examination table to improve scan quality for larger breasts; upgraded antennas to improve imaging of the posterior of the breast; integrated thermoregulation; chemical stabilization of the transition liquid; integration of a patient positioning aid module in the MBI scanner; enhanced radiofrequency emission/reception chain; and mechanical support of the sensor array to improve signal stability and scan repeatability.
The researchers tell Physics World that a second clinical investigation is planned for early 2022 using the advanced prototype. “A multi-stage adaptive design is being considered for implementation in this clinical study, allowing us to first assess the technical performance of the upgraded Wavelia MBI prototype on an initial patient dataset,” they explain. “The identification of clinical cases for which Wavelia MBI could bring added value, as an adjunct imaging modality to conventional imaging, will be ultimately explored on larger and more diverse patient datasets.”
There is still much work to be done to make MBI a feasible breast cancer scanning technology. In addition to reducing the exam time, which for this study averaged 50 min, MBI systems must be able to image a wide range of breast sizes, detect small, non-palpable breast pathologies and achieve a non-negligible false positive rate. Large MBI clinical trials are required to confirm automated and consistent detection of various breast pathologies in breasts of differing tissue density.
The researchers, however, are positive about the technique’s capabilities. “The Wavelia MBI system holds significant potential for detecting breast abnormalities, while offering the patient a favourable experience over conventional mammography. This novel modality may also add significant value to the existing detection paradigm for ILC, which can evade conventional imaging,” they conclude.