
Optogenetics, in which neurons are engineered to be activated or inhibited by light, provides valuable insights into underlying mechanisms of brain function and holds promise for the treatment of neurological disorders. However, the blue-green wavelengths required to turn neurons on or off scatters strongly and cannot penetrate deep into the brain, necessitating the use of fibre probes to deliver the light.
To avoid this invasive approach, a research team headed up at the RIKEN Brain Science Institute is investigating the use of upconversion nanoparticles (UCNPs) to enable delivery of laser light from outside the skull. These UCNPs absorb near-infrared laser light, which can penetrate deeper into brain tissue, and emit the blue-green photons required for neural stimulation (Science 359 679).
“Optogenetics has been a revolutionary tool for controlling neurons in the lab, and hopefully someday in the clinic,” said research group leader Thomas McHugh. “Unfortunately, delivering light within brain tissue requires invasive optical fibres. Nanoparticles effectively extend the reach of our lasers, enabling the ‘remote’ delivery of light and potentially leading to non-invasive therapies.”
In tests in mice, the researchers demonstrated that the UCNPs could serve as optogenetic actuators of transcranial NIR light to turn on neurons in various brain areas. Electron microscopy showed that UCNPs injected into mouse brains remained localized in the injection area.
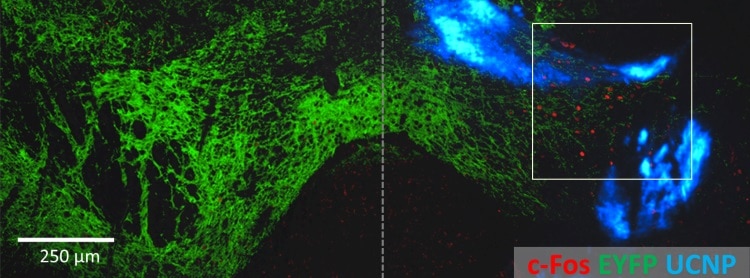
“The nanoparticles appear to be quite stable and biocompatible, making them viable for long-term use,” said McHugh. “Plus, the low dispersion means we can target neurons very specifically.”
In addition to activating neurons, the UCNPs could also be used for inhibition, for example to silence seizures. The researchers injected nanoparticles tuned to emit green light into the hippocampus and energized them with laser pulses at the surface of the skull. Hyperexcitable neurons were effectively silenced in these mice.
In another brain area, the medial septum, nanoparticle-emitted light contributed to synchronizing neurons in an important brain wave called the theta cycle. And in mice with learned fear memories, the freezing behaviour associated with these experiences was evoked by blue light-emitting UCNPs, also in the hippocampus.
These neural activation, inhibition and memory recall effects were only observed in mice that received nanoparticle-mediated optogenetic stimulation, not in control animals that received laser light without the UCNP injection.
The nanoparticles are compatible with various light-activated channels currently used in optogenetics and can be employed for neural activation or inhibition in many deep-brain structures. The authors note that the technique could, one day, complement or extend current approaches to deep-brain stimulation and therapies for neurological disorders in humans.



