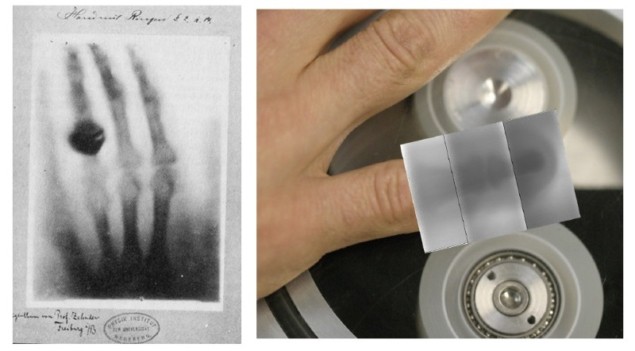
History has seen many notable innovations in the field of medical X-ray imaging. From Wilhelm Conrad Röntgen’s discovery of X-rays in 1895 and Godfrey Hounsfield’s development of CT in 1967, both of which led to the award of Nobel prizes, through to the introduction of dual-energy X-ray absorptiometry in 1971.
But 1971 is a long time ago; perhaps we are overdue for the next disruptive innovation? According to Peter Seitz, from Hamamatsu Photonics Europe, inspiration for new X-ray imaging modalities may come from techniques currently being developed for photonics applications. “I will show five exciting new technologies that could lead to disruptive new medical imaging modalities,” he told the audience at the recent MediSens conference in London.
TOF range imaging
The first inspiration, said Seitz, is optical time-of-flight (TOF) range imaging. This is the technology employed in driverless cars to sense distances to nearby objects, and can also be found in many smartphones for proximity sensing.
TOF range sensing uses the travel time of reflected light to measure distances, and requires nanosecond pulse generation and detection. For optical systems, this is enabled by the use of vertical-cavity surface-emitting lasers (VCSELs) and single-photon avalanche diode (SPAD) photodetectors. But can this be achieved using X-rays?
X-rays are conventionally created using a radioactive element or an X-ray tube. The former requires a shutter to create pulses and cannot provide nanosecond switching. X-ray tubes, meanwhile, can only be switched at 100 ms speeds. Seitz suggested a third option: cold-catheter electron emitters. These miniaturized X-ray tubes are based on carbon nanotubes and can switch at rates of less than 10 ps, creating ultrashort X-ray pulses. He noted that fast detectors with 100 ps resolution also exist.
“TOF X-ray imaging may be around the corner; we have the sources and they are not expensive,” said Seitz. “Perhaps TOF imaging is not only for cars, but also for clinical applications.”
Direct photon detection
Next, Seitz discussed use of perovskite semiconductors for direct detection of visible photons. Perovskites, materials with the same crystal structure as the perovskite mineral CaTiO3, have recently been used to create quantum dots (QDs). Such perovskite QDs can be used to create high-quantum-efficiency light emitters and detectors with tailorable wavelengths.
Direct detection of X-rays remains challenging, however. Silicon detectors are not suitable for X-ray detection, while the more optimal detector – cadmium telluride – is very expensive. So can we use perovskites for direct X-ray photosensing? It appears so, said Seitz.
He explained that it is possible to grow high-quality X-ray detectors based on lead-halide perovskite crystals. Detector crystals with dimensions of 2–10 mm exhibit almost identical absorption properties to cadmium telluride, but at a cost of approximately one Euro per crystal.
“So are we there already? Not quite,” Seitz explained. “The problem is that everything works but it is not stable, something migrates in the perovskite. We need to fix this… then there will be sleepless nights for the cadmium telluride guys! Watch this space.”
Phase contrast imaging
Detecting cancer using X-ray absorption imaging is hindered by the fact that the tumour has the same absorption properties as surrounding tissue. The two tissues do, however, have different refractive indices. Inspired by dark-field optical imaging using phase contrast techniques, Seitz’s third suggestion was X-ray phase contrast imaging.
In optical phase contrast techniques, a small difference in refractive index causes light to be slightly deflected. By imaging this refracted light, it’s possible to distinguish structures of similar transparency and visualize far more detail. This same approach can be applied for X-ray imaging.
Phase-contrast X-ray imaging exploits refraction and interference effects to create images with significantly higher contrast than in conventional X-ray radiography, and can reveal boundaries between materials with differing refractive indices. Using a standard X-ray source, phase-contrast X-ray imaging allows simultaneous detection of a conventional absorption radiograph, a differential phase image and a scattering image.
“This is here – not yet in production, but it has been demonstrated, it’s possible, and it may come soon,” Seitz told the audience.
Content-sensitive spectroscopy
Another recent development in smartphone technology is the incorporation of optical spectroscopy. Phones with built-in spectrometers could be used to sense the environment, to test air quality, for example. “Spectroscopy is next big thing in smartphones,” said Seitz. “Can I have this for X-rays?”
X-rays used in medical applications interact with tissue via either the photoelectric effect or the Compton effect. Does this mean that it’s only possible to create two different images? Not if you can detect individual X-ray photons, Seitz explained. Then it should be possible to measure the energy of each photon and deduce the elemental composition of a target from its absorption spectrum.
“It is possible to do X-ray spectroscopy, provided you have detectors and stable sources and can reliably say for each photon when it arrived at the detector,” said Seitz. He described an experiment employing element-sensitive X-ray radiography to determine the relative content of two materials in a composite sample.
Triboluminescence
The last inspiration on Seitz’s list was triboluminescence – light generated when chemical bonds are broken in a material subjected to friction, impact or breakage. For example, flashes of blue light are produced when crushing a sugar cube, or unrolling adhesive tape.
It has also been shown that, in a vacuum, X-ray flashes with energies of up to 100 keV can be produced simply by unrolling Scotch tape. What’s more, researchers have already demonstrated that such triboluminescence can be used to create X-ray images of a finger, using a dental detector.
“The result is reminiscent of the first image taken by Röntgen,” noted Seitz.