The shape of a nanoparticle is important for how easily it can penetrate into a tumour, with rods and hollow cubes entering more readily than discs and spheres. This new result, from researchers at Georgia Institute of Technology and Washington University Medical School in the US, could lead to better nanoparticle-based cancer diagnosis and therapy.
Cancer drugs and markers can be delivered to the inside of tumour cells by encapsulating the pharmaceuticals within tiny nanoparticles about 100 nm in size. The idea is to coat the nanoparticle with molecules that have an affinity for the target tumour. Only when the nanoparticle enters the tumour is its pharmaceutical cargo released, thereby minimizing side effects and improving marker accuracy. The efficacy of this process depends on a variety of factors, and one of the most critical of these is the shape of the structure. It is therefore important to understand which nanoparticle shapes are best, but such studies are complicated because it is difficult to track the movement of nanostructures in the body.
Four distinct shapes
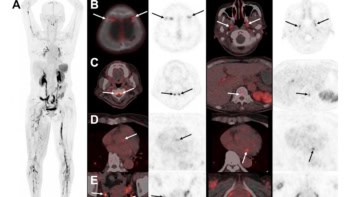
Now, a team of researchers led by Younan Xia at Georgia Institute of Technology and Yongjian Liu of Washington University has solved this problem using gold nanostructures that contain radioactive gold-198 in their crystal lattices. The nanostructures were made in four distinct shapes – solid discs, rods, spheres and hollow cubes – and were all roughly the same size (between 50 and 100 nm across). By using an external detector to measure gamma rays from the gold-198, the team could follow the nanostructures as they distributed themselves in live mice with tumours.
The researchers found that the different shapes entered the tumours at different rates and also distributed themselves differently. “Our findings should help us develop better strategies for cancer imaging and therapy,” explains Xia.
The study reveals that the rods (10 nm in diameter and 50 nm long) did not accumulate in the tumour sites as readily as the other shapes. However, those rods that did arrive at the surface of the tumour were better at penetrating into its core. In contrast, the spheres (about 50 nm in diameter) and discs (7 nm thick and 100 nm in diameter) were only able to reside on the surface of the tumours.
Shape matters
The cubes – which were hollow “nanocages” with an edge length of about 50 nm – were also able to penetrate to the core of the tumours. Xia believes that the cubes were able to reach the tumour cores because of their hollow structure and relatively low density. “Our study shows that the shape of gold nanostructures is important for how these particles enter and distribute themselves in tumours,” concludes Xia.
The team says that it will now focus on reducing the size of the nanostructures it studied so that these can stay in the bloodstream for longer periods and also better penetrate cancer cells. “We also plan to test these radioactive nanostructures on other tumour models, such as orthotopic mouse breast-cancer cells,” says Xia. “We will further functionalize the surface of these structures with targeting ligands so that tumour cells take them up more readily.”
The research is described in ACS Nano.
- This article first appeared on nanotechweb.org