Changes in the strength between synapses thought to be linked to memory might be more “digital” than previously thought, according to new work by researchers at Thomas Jefferson University in the US. The unexpected result suggests that synaptic plasticity might work by the addition of small same-size modular units of proteins, dubbed nanomodules, rather than by the synaptic connections simply becoming bigger – as was observed before.
Synapses are the biological junctions between neurons and they transform a voltage spike (action potential) arriving from a pre-synaptic neuron into a discharge of chemical neurotransmitters that are then detected by a post-synaptic neuron. These are then transformed into new spikes, leading to a succession of pulses that either become larger or smaller. This fundamental property of synaptic behaviour is known as short-term plasticity, which is related to a neural network’s ability to learn
Unexpected structural changes in spines
As the network learns, the connections between neurons become stronger and this leads to long-lasting changes in the size of the membranous protrusions, or spines, that protrude from neuronal dendrites. These spines, which nearly touch at the synapse, comprise a long thin neck and a rounder swollen head.
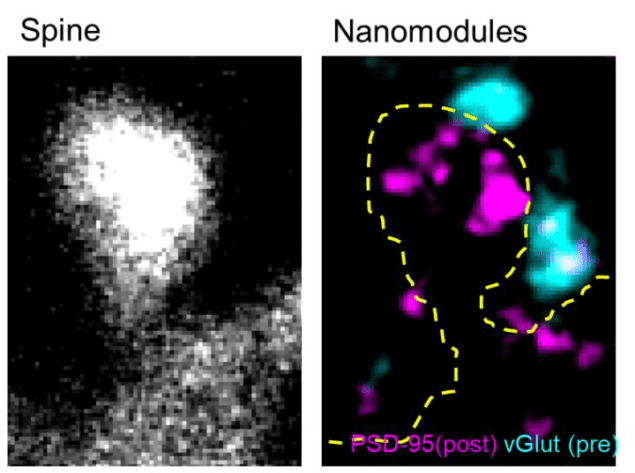
To better understand the molecular changes at play in neurons during learning, researchers led by Matthew Dalva employed a technique called multicolour stimulated emission depletion microscopy (STED) to study the connections between neurons that strengthen. Thanks to their images, the researchers have discovered hitherto unseen structural changes in the spines as they become bigger during learning.
Two and three colour STED
“Our technique relies on two and three colour STED to image how key synaptic proteins are organized on the nanoscale,” explains Dalva. “We were able to combine this approach with confocal imaging to examine the relationship between the morphology, or shape, of the dendritic spines and how the proteins that regulate spine function are organized.”
As well as this work, which was done on fixed cells, the researchers also conducted two-colour STED combined with confocal imaging to visualize what happens at spines in living neurons. “In these experiments, we induced spine plasticity (that is, we made the spine get bigger) and watched how the nanostructures at the synapses changed.”
Nanomodules begin to jiggle and move
Team member Martin Hruska’s experiments revealed that as spines increase in size, they add modular units of synaptic proteins, dubbed nanomodules. “This result suggests that changes in synaptic strength – changes that scientist think are linked to memory – might be more digital than previously thought,” Dalva tells nanotechweb.org. “The finding, which will need to be confirmed in future work, suggests that synaptic plasticity might work by adding small same-size units of proteins rather than the synaptic connections themselves simply scaling up in a linear fashion.”
And that was not all: “we also found that the nanomodules begin to jiggle and move around the synaptic spine, with pre- and post-synaptic connections always in lock step,” says Hruska.
Many new questions to be answered
The research, which is detailed in Nature Neuroscience doi:10.1038/s41593-018-0138-9, raises many interesting questions, adds Dalva. “In this work, we only looked a few different types of synaptic proteins, for instance. Are others also organized in the same way? How are the nanomodules we observed generated, and how do they stay aligned across the synaptic cleft between neurons? And how do the nanomodules increase in number?
“Could this work help us better understand how synapses work? For example, does each nanomodule operate independently or are they somehow functionally linked? These are all key questions we plan to examine next.
“Finally, our observations could also help us better understand the maladaptive learning that occurs in disorders such as addiction or other neurological diseases, in which abnormally strong or weak connections between neurons are thought to be responsible,” he adds. “It is yet difficult to say how the nanomodules behave in disease states, but our work offers a new way to explore those questions.”



