A new method for imaging the flow of blood has been developed by researchers in the US. By using ultrasound to thermally tag blood, along with photoacoustics to image the resulting heat flow, the new technique is considerably more sensitive than the conventional Doppler ultrasound method that is currently used. While presently at the in vitro testing stage, this technique might have a variety of clinical applications, especially in medical diagnosis.
Being able to image the flow of blood within deep tissue would provide valuable information for the diagnosis and understanding of many diseases, with potential applications including functional brain imaging, detection of vascular diseases (such as atherosclerosis – the thickening of artery walls with calcium and fat) and the analysis of blood within tumour microenvironments (commonly having complicated vasculatures with intermittent blood flows), which could help in the early detection of cancer.
Going with the slow flow
Currently, flow imaging is undertaken through Doppler ultrasound (or sonography) in which the flowing blood causes a Doppler shift in the reflected ultrasound waves that can be measured and used to discern the flow rate. However, this technique has its limitations. Doppler ultrasound has poor sensitivity to blood, which weakly scatters in comparison with tissue. This issue is even more pronounced at the low frequencies that are commonly used to probe deeper into flesh. At slow flow rates, the small Doppler shifts from the moving blood can therefore be difficult to distinguish from shifts that are caused by the surrounding tissues. Indeed, the effect is undetectable for blood flowing slower than about 10 mm s–1.
One possible alternative to Doppler ultrasound is photoacoustic imaging, which uses short pulses of low-energy laser light to locally heat the target for analysis. This heating results in thermal expansion, creating ultrasound waves in proportion to the optical absorption of the target tissue. When recorded, this emitted ultrasound can therefore be used to create a map of the target’s absorptivity. While this method works well at shallow tissue depths (up to 1 mm deep), flow sensing in deep tissue is hindered by the high densities of blood cells.
Thermal tagging
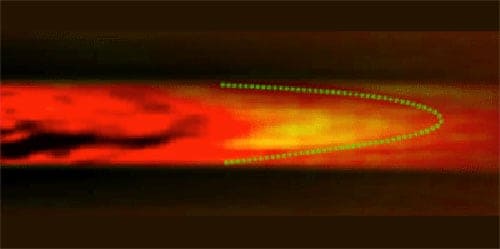
Photoacoustic imaging is, however, very sensitive to temperature variations. Taking advantage of this, Lidai Wang and colleagues at the Washington University in St Louis have developed an alternative method of analysing blood flow by combining photoacoustic imaging with thermal ultrasound tagging that they refer to as “photoacoustic flowgraphy”. The researchers used focused ultrasound to locally heat a fixed part of a blood vessel and mapped out the resulting temperature distribution along the vessel as the blood flowed downstream.
As the laser pulse expands the material, it generates sound waves of a particular amplitude that, in turn, vary with the temperature. In a test set-up, the team could see how the heated sample moved by tagging the sample with the laser 10 times a second. Comparing subsequent images of the sound waves (taken using an array of detectors) allowed the researchers to follow the flow in a 1.5-mm tube of cow’s blood, including its faster speed at the centre of the tube.
“The spatial frequency (or period) of the temperature distribution changes with blood-flow speed,” explains Wang, with blood that flows faster generating a longer spatial period and a lower spatial frequency. This enables the flow speed to be calculated from the spatial frequency of the photoacoustic images recorded. Compared with traditional Doppler sonography, photoacoustic flowgraphy is considerably more sensitive, enabling the team to measure capillary-level blood flow at speeds as low as 0.24 mm s–1. “[This] is four times slower than Doppler sonography,” Wang adds. Furthermore, use of spectral photoacoustic imaging with this new method could provide other useful functional information, such as the blood’s oxygen saturation and even the metabolic rate of oxygen.
Assessment tool
“What I find particularly exciting is how this can be combined with functional and anatomical photoacoustic imaging to determine several tumour properties at once using one measurement,” comments Eric Strohm, a biophysicist at Ryerson University in Canada who was not involved in this study. “While in the early stages of development, this technology could eventually see clinical applications as an early assessment tool for evaluating tumours.”
The team is now looking at improving the technique by developing a reflection-mode system in which the ultrasound heating and photoacoustic transducers would be located on the same side of the subject tissue. Paving the way for in vivo testing, this set-up would ultimately allow access to a wider variety of anatomical sites for clinical applications.
The research is published in Physical Review Letters.