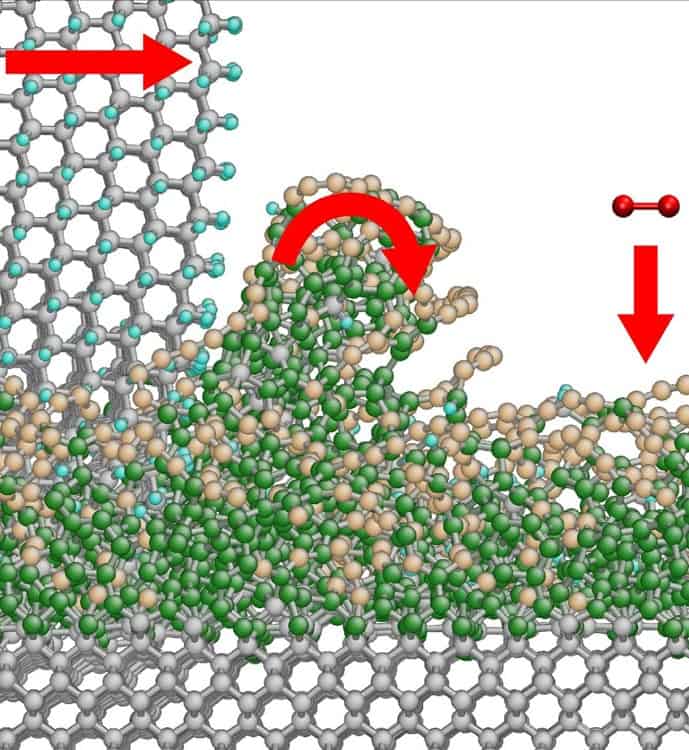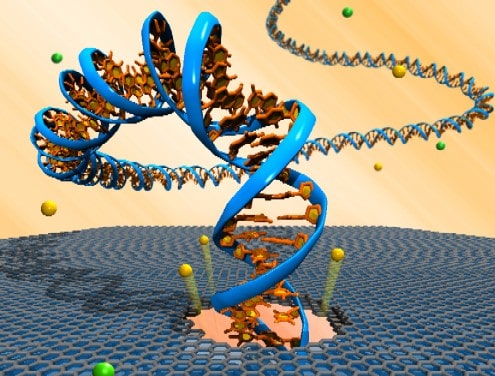
DNA being pulled through a sheet of graphene. Courtesy: Robert R Johnson
By James Dacey
High achievers who commit their working life to fundamental research do not tend to be driven primarily by money. But the chance to win a slice of a $10 million prize must at least bring a bounce to the step of a scientist as they whizz around the lab. That’s the situation for researchers developing DNA sequencing technologies who stand a chance of sharing the Archon Genomics X Prize, which will pay this money to the first privately funded company that can accurately sequence 100 human genomes in 10 days.
Earlier this week, researchers at the University of California reported an important breakthrough in one of the promising techniques that could scoop this prize. Kate Lieberman and her colleagues are seeking to develop a system known as nanopore sequencing. The basic idea is that by feeding DNA through pores in thin films that are so small that the molecules almost fill the gap, the DNA could alter the electronic properties of the film. If researchers can also develop a highly sensitive way of monitoring electronic currents passing across the gap, they could in theory identify individual bases – A, C, G and T.
The idea was first mooted in the mid 1990s, but naturally there have been a number of challenges along the way. Lieberman and her colleagues have addressed one of these, which is to find a controlled way of passing DNA strands through the gap. They manage to pass a single strand of DNA through a nanopore in protein by coupling the DNA with a polymerase enzyme, which can then pass smoothly through the gap and be detected in the presence of an electric field.
The researchers, who report their findings this week in Journal of the American Chemical Society, intend to develop their technology by working with their industrial partners, the UK-based company Oxford Nanopore. “The ‘strand sequencing’ method of DNA sequencing using a nanopore has been studied for many years, but this paper shows for the first time that DNA can be translocated by an enzyme using methods that are consistent with a high throughput electronic technology,” said Gordon Sanghera, CEO of Oxford Nanopore.
If you are a member of the Institute of Physics, you can read more about nanopore sequencing and the incentive of the X Prize in this recent feature article from the print edition of Physics World.