
Primary-school children, and the rest of us too, are continuously being showered by unseen muons, a heavier relative of the more familiar electron. These muons are created when energetic cosmic rays, including protons and alpha particles, hit our atmosphere and produce a shower of particles as they slow down. At sea level, muons arrive at a rate of about one per square centi-metre per minute.
Muons remain unnoticed unless you have the right equipment to look for them. Several years ago I was therefore excited to read about a successful US-based outreach project called Cosmic Watch. Started by particle physicists, it allows members of the public to make muon detectors for less than $100 and observe these tiny particles for themselves. At the heart of the detector is a silicon photomultiplier chip, which measures the few blue photons emitted by a plastic scintillator whenever a muon passes through.
Inspired, I started to build muon detectors based on the Cosmic Watch design. But when I had got one working, I needed something to do with it. A work trip to Belgium on the Eurostar train presented one such opportunity. I took the detector with me (curiously, no questions were asked at security) and, sure enough, as we travelled through the Channel Tunnel between Britain and France, it recorded a lower rate of muons than at sea level. The sea and seabed were shielding the detector.

As fun as that was, muonic measurements are better shared. So, along with another physicist parent, Lisa Ibberson, I got in touch with Kate Cooke, who teaches science at Coton Church of England Primary School in Cambridgeshire, where my son attends. Together, the three of us applied for money from the Institute of Physics School Grants Scheme. Our idea was to work with pupils to teach them about muons, get them to design a muon detector and finally install it in the school.
In July 2019 we were delighted to hear that our grant was successful and the real work began. We held an initial assembly at school that October using a water pistol and a few slides to introduce ourselves and muons. With the pupils in years 3 and 4 (ages 7–9), we drew pictures of the cascading particles resulting from a cosmic-ray air shower. Meanwhile, the children in years 4 and 5 (ages 8–10) were in charge of how the detector looked.
Together we decided that it should have a muon counter and a display that flashed different colours, depending on the muon’s energy. We jointly defined various parameters of the display, including its size, the colours of the flashes and the number of digits on the counter. Finally, we talked with year-6 pupils about the data the detector would produce, with the help of some edible Smartie bar graphs of course. And behind the scenes we were busy ordering printed circuit boards, soldering components and programming microcontrollers.
In January 2020 we returned to the year 4/5 class with a red flashing prototype encased in a shoe box. We got some great feedback. The colour red was no good – it was too much like a warning light and too bright. We therefore dimmed the display and democratically chose blue, green and amber for the colours. The pupils also told us we needed a switch to turn off the display when the flashing got too distracting. Finally, we decided to have eight digits in the detector so it could count to 99,999,999 muons – over roughly the time pupils spend in school (at a count every two seconds we were expecting about 15 million counts per year).

Unfortunately, when the pandemic struck we couldn’t continue to work directly with the children. Instead, over the summer I worked at home, quietly improving the detector’s electronics. In fact, on walks through the village where I live, people would often ask me what had happened to the muon project. I’d tell them we’d get back to it when we can and fortunately, by September 2020, schools re-opened and we started to think about the final switch-on.
Originally we envisaged a school assembly with a rowdy group countdown to the detector being turned on. We couldn’t do that with social-distancing measures in place, so instead planned a virtual switch-on for December 2020 with pupils from years 4 to 6 in their classrooms, Cooke at school, and me and Ibberson joining from our homes. And so, after revving the pupils up with a quiz to remind them about muons, we turned the device on.
How we can widen career aspirations for the next generation
It worked, phew! We then spent a few minutes watching the detector count up to 60, before opening the floor to questions. And, wow, what great questions they asked. How can a muon travel through 10 metres of concrete? Why do they decay into electrons? If the detector had a bigger area, would it count more muons? Has a detector like this ever been made before? Where do the cosmic rays that generate the muons come from?
Overall, the project was a great success. We had wonderful engagement from the pupils, who had contributed to the design of a fun scientific instrument. In fact, the switch-on of the detector, though virtual, was every bit as exciting as if it had happened in school. Looking to the future, we hope that this strange machine from a strange year counts up to 99,999,999 many times over its lifetime – and that it continues to provoke curiosity from primary-school pupils in Coton for years to come.


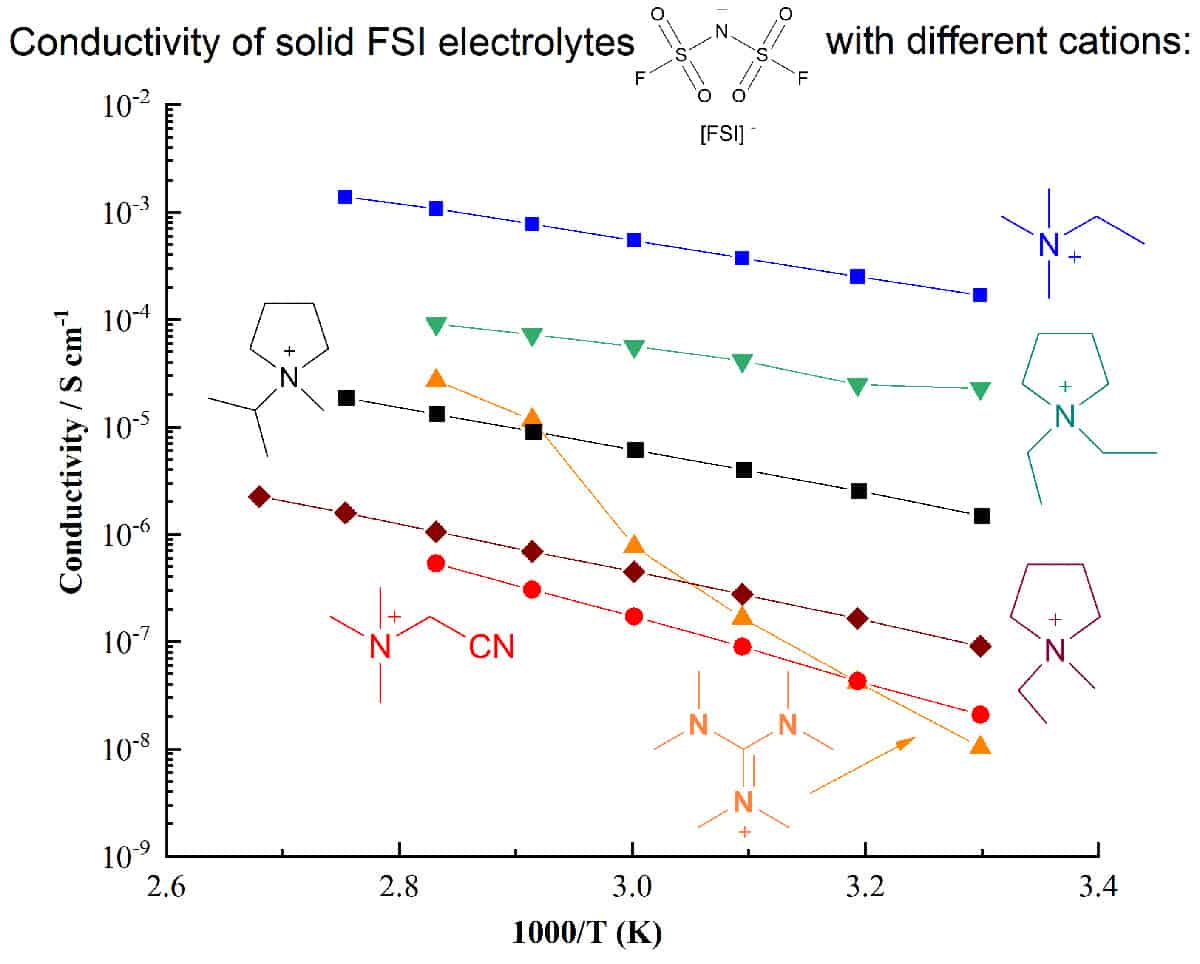 Electrolyte development is a critical component in the quest for higher-performing energy storage devices. Ionic electrolytes such as ionic liquids, plastic crystals and their polymer composites can offer important safety and performance advantages over traditional molecular-solvent based systems, particularly for devices utilizing reactive metals such as lithium or sodium.
Electrolyte development is a critical component in the quest for higher-performing energy storage devices. Ionic electrolytes such as ionic liquids, plastic crystals and their polymer composites can offer important safety and performance advantages over traditional molecular-solvent based systems, particularly for devices utilizing reactive metals such as lithium or sodium. Prof. Jenny Pringle works at the Institute for Frontier Materials at Deakin University, Australia. She is a chief investigator in the ARC Centre of Excellence for Electromaterials Science (ACES) and the ARC Industrial Transformation Training Centre “StorEnergy”. She received her degree and PhD at The University of Edinburgh in Scotland, UK, before moving to Monash University, Australia, in 2002. From 2008–2012 she held an ARC QEII Fellowship, investigating the use of ionic electrolytes for dye-sensitized solar cells. Pringle moved to Deakin University in 2013. There she leads research into the development of new ionic liquids and organic ionic plastic crystals for applications including thermal energy harvesting, gas separation membranes, and lithium and sodium batteries.
Prof. Jenny Pringle works at the Institute for Frontier Materials at Deakin University, Australia. She is a chief investigator in the ARC Centre of Excellence for Electromaterials Science (ACES) and the ARC Industrial Transformation Training Centre “StorEnergy”. She received her degree and PhD at The University of Edinburgh in Scotland, UK, before moving to Monash University, Australia, in 2002. From 2008–2012 she held an ARC QEII Fellowship, investigating the use of ionic electrolytes for dye-sensitized solar cells. Pringle moved to Deakin University in 2013. There she leads research into the development of new ionic liquids and organic ionic plastic crystals for applications including thermal energy harvesting, gas separation membranes, and lithium and sodium batteries.



 Thomas Dieing is technical product manager for the WITec alpha300 product line and its accessories. He obtained his PhD from La Trobe University, Melbourne, Australia, in 2005 investigating the MBE growth of nitrogen containing III/V semiconductors. In 2006 he joined WITec’s application team and became director of applications and support. In his role as product manager since 2019 he is responsible for all activities related to the product development process.
Thomas Dieing is technical product manager for the WITec alpha300 product line and its accessories. He obtained his PhD from La Trobe University, Melbourne, Australia, in 2005 investigating the MBE growth of nitrogen containing III/V semiconductors. In 2006 he joined WITec’s application team and became director of applications and support. In his role as product manager since 2019 he is responsible for all activities related to the product development process. Ute Schmidt studied physics at the Babes Bolyai University in Cluj-Napoca, Romania, and obtained her PhD from the University of Karlsruhe, Germany. Through her work with in situ scanning tunneling microscopy in an electrochemical environment, she was introduced to scanning probe microscopy. During her postdoctoral scholar position at Karlsruhe and North Carolina State University in Raleigh, USA, she continued to work with metal deposition on different substrates with STM and Atomic Force Microscopy. Schmidt worked for six years as a project manager at Molecular Imaging Corp., Phoenix, USA. She has been an applications manager at WITec since 2003.
Ute Schmidt studied physics at the Babes Bolyai University in Cluj-Napoca, Romania, and obtained her PhD from the University of Karlsruhe, Germany. Through her work with in situ scanning tunneling microscopy in an electrochemical environment, she was introduced to scanning probe microscopy. During her postdoctoral scholar position at Karlsruhe and North Carolina State University in Raleigh, USA, she continued to work with metal deposition on different substrates with STM and Atomic Force Microscopy. Schmidt worked for six years as a project manager at Molecular Imaging Corp., Phoenix, USA. She has been an applications manager at WITec since 2003.