3D bioprinting has the potential to revolutionize medicine because it will allow patient-specific human tissues to be fabricated in the lab. The technology relies on the availability good “bioinks” – biomaterials laden with cells that are used to print the biological structure – but evaluating the printability of individual inks in experiments is proving to be time-consuming, costly and complicated. Researchers are therefore turning to computer simulations as a complementary technique.
“As well as allowing us to evaluate the printability of a bioink, simulations could also help us choose the printing technique that should be employed depending on the target tissue,” explains Kajsa Markstedt, who is at the Wallenberg Wood Science Center (WWSC) in Gothenburg, Sweden. “Such techniques vary depending on the viscosity and nature of the ink being printed, and include ink-jet printing, laser-induced forward transfer, microvalve- and extrusion-based bioprinting.”
“This provides a better understanding of why certain printer settings and bioinks work better than others”
Kajsa Markstedt, Wallenberg Wood Science Center
Markstedt and colleagues worked with Johan Göhl’s team at the Fraunhofer Chalmers Centre to model the process using a computational fluid dynamics tool (Biofabrication 10 034105). “To model how a bioink is dispensed, we used its mass flow rate and density as input in our calculations,” says Markstedt. “These parameters are the ones most commonly evaluated in experiments when printing designs such as lines, grids or cylinders.”
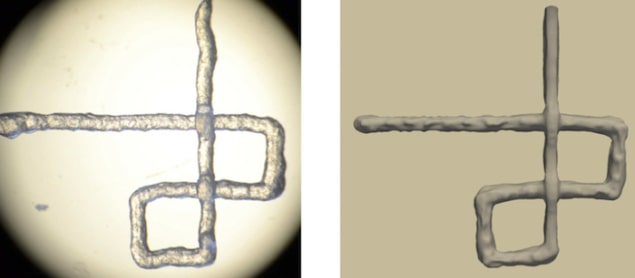
Markstedt explains that the printability is usually determined with regards to the ratio of line width to nozzle diameter, the number of layers that can be printed before the structure collapses or the curvature of printed lines. “In our simulations, we also used the printing path of a grid structure as input.”
The Fraunhofer Chalmers Centre scientists also modelled the wettability of bioinks on a substrate using a dynamic contact-angle model that depends on surface tension and an apparent contact angle as input. Their full rheology model was based on ink-oscillation frequency data obtained in experiments, and the viscoelastic behaviour of the material.
Model mirrors reality
Simulations on cellulose nanofibril (CNF) inks produced very similar outcomes to experimental results obtained in laboratory evaluations, even for bioinks with very different rheological properties. What’s more, the computer model enabled the researchers to follow the printing process in real time, and study the behaviour of different inks while they are being dispensed.
“In experimental evaluations, we often only have the properties of the final, printed grid structure to go on,” explains Markstedt. “This is a time-consuming way to develop new bioinks or to optimize printing parameters for a specific ink. It is also expensive since the prepared bioink containing cells is precious.”
The biostructure also needs to be tested shortly after being printed – while the cells are still viable – which limits the time for extensive evaluations. “This often leads to many bioinks being printed at printing parameters that have not been optimized for a specific bioink composition,” continues Markstedt. “The result is that the right architecture is not produced, which can be catastrophic because the printed tissue does not function properly. For example, the printed line may be too thin causing the structure to break, or too thick, which prevents nutrients and oxygen reaching all the cells in the bioink.”
The teams is confident that the new simulation tool overcomes all these problems, while also providing much more feedback during the printing process itself, such as how viscoelastic- and shear stresses are distributed in the ink. “This provides a better understanding of why certain printer settings and bioinks work better than others,” says Markstedt. “For example, it allows us to isolate individual parameters, such as printing speed, printer nozzle height, ink flow rate and printing path to study how they influence printing.”
Looking forward, the researchers are now planning to expand their studies to model the flow of ink inside pre-defined nozzle geometries. “This addition to the model will allow us to observe what effect shear stresses from the nozzle have on the printing process,” says Göhl. “This will help us to determine how different printing pressures and nozzle shapes affect the bioprintability of a bioink.”
- Read our special collection “Frontiers in biofabrication” to learn more about the latest advances in tissue engineering. This article is one of a series of reports highlighting high-impact research published in Biofabrication.



