The best way to predict a good outcome for paediatric patients with osteosarcoma is by performing FDG-PET/CT scans at regular intervals in the first several months after the start of chemotherapy, according to a study published in the January issue of the Journal of Nuclear Medicine.
Researchers from several children’s hospitals and cancer research facilities found that routine images at weeks 5 and 10 and the percentage change in the maximum standardized uptake value (SUVmax) from baseline to week 10 were predictors of a favourable response to standard chemotherapy in combination with bevacizumab. The findings suggest that early changes in treatment regimens before surgical intervention could result in more favourable outcomes for patients.

“This well-organized, clinical therapeutic study allowed us the opportunity to assess the usefulness of FDG-PET/CT imaging in patients with a single uncommon disease treated uniformly on a rigorous chemotherapy protocol and evaluated comprehensively at prospectively identified milestones during therapy,” said study co-author Barry Shulkin from the department of radiological sciences at St. Jude Children’s Research Hospital in Memphis, TN (J. Nucl. Med. 59 25).
Osteosarcoma occurrences
While osteosarcoma is the most common primary bone malignancy in children, the disease is quite rare and accounts for only 0.1% of all tumours, according to one previous study. With curative options that include chemotherapy and surgical resection, the chances for long-term survival for these paediatric patients has improved.
As background to this study, the researchers, led by Najat Daw, a paediatric oncologist at MD Anderson Cancer Center in Houston, have been developing a new chemotherapeutic approach to osteosarcoma, incorporating the antiangiogenic agent bevacizumab, which has been used for other paediatric cancers.
“Our molecular imaging group has been particularly interested in evaluating the utility of metabolic imaging in multiple tumour types in children, adolescents and young adults,” Shulkin wrote in an email to AuntMinnie.com. “The challenge was to identify a role for FDG-PET/CT imaging in the evaluation and management of patients with osteosarcoma, if one exists.”
In the study, the researchers prospectively enrolled 34 consecutive patients (median age, 12.2 years; range, 6.8-19.1 years) with newly diagnosed, high-grade, biopsy-proven localized or metastatic osteosarcoma between June 2008 and May 2012.
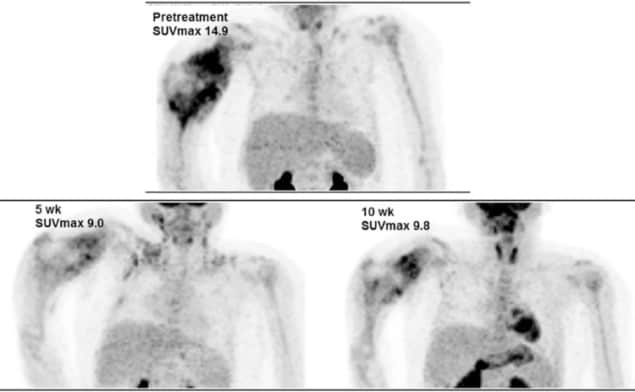
The 17 male and 17 female subjects initially underwent evaluation with chest CT, MRI and X-rays of the tumour bed. In addition, whole-body FDG-PET/CT (Discovery LightSpeed, GE Healthcare) was performed to create a baseline, with follow-up scans at five and 10 weeks after the start of chemotherapy. Primary tumours were then resected during the 10th week.
Chemotherapy included cisplatin and doxorubicin at the start of the sessions and five weeks later, along with high-dose methotrexate at the third, fourth, eighth and ninth weeks. Bevacizumab was administered three days before the first dose of chemotherapy and then on the first day of the third and fifth weeks of chemotherapy.
The most common locations for the primary tumours were the femur (17 cases, 50%), tibia (nine cases, 26%), and humerus (five cases, 15%). Twenty-five patients (74%) had nonmetastatic osteosarcoma, while the other nine (26%) had metastatic disease at diagnosis.
SUVmax stats
SUVmax proved to be a significant predictor of outcomes at week 5 after the start of chemotherapy and again at week 10. At baseline, SUVmax, however, was not a contributing factor to a patient’s outcome. Paediatric patients with a higher SUVmax at week 5 or week 10 were less likely to respond favourably to chemotherapy.
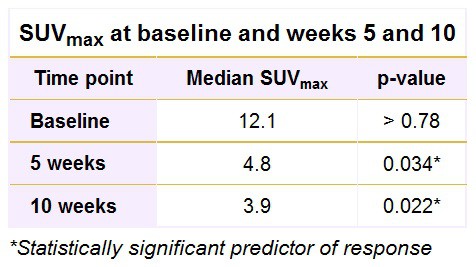
Consequently, the percentage change in SUVmax from baseline to week 5 (60%, p < 0.001) and from baseline to week 10 (68%, p = 0.021) both significantly predicted a response to treatment. Paediatric patients with a larger percentage decrease in SUVmax were more likely to respond to treatment.
The researchers also calculated receiver operating characteristic (ROC) curves for SUVmax at five and 10 weeks and based on the percentage change from baseline to week 10. Optimum cutoff points for SUVmax were 4.04 at week 5 and 3.15 at week 10, along with a 60.24% decrease from baseline at week 10. Based on those values, the SUVmax parameters achieved high sensitivity.

As for overall survival at a median follow-up time of 3.6 years, 24 (70%) of the 34 patients were still alive. Sixteen patients (47%) experienced adverse events, which included disease relapse, progression, a second malignancy or death from any cause.
Event-free survival
The researchers did not find statistically significant associations between event-free survival and SUVmax at baseline, week 5 (p = 0.11), or week 10 (p = 0.12). There also was no significant association between event-free survival and changes in SUVmax from baseline to weeks 5 or 10 or between event-free survival and percentage changes in SUVmax from baseline to weeks 5 or 10.
“The reason may be the small sample size; statistical significance may become apparent with larger numbers of subjects,” the authors wrote.
Based on the influence of SUVmax and the timing of FDG-PET/CT scans, Shulkin and colleagues are developing a new protocol for the treatment of osteosarcoma. “We intend to perform FDG-PET/CT imaging earlier than in the previous study to further explore the time course of chemotherapy in relation to cell killing,” he said.
The researchers also plan to work with colleagues from the University of Wisconsin to perform a radiomic analysis of the scans.
 “We want to determine if nonstandard analyses requiring high computational capacities will reveal additional information about these tumours and their response to therapy,” Shulkin said. “In addition, we are planning to evaluate other radiotracers, principally carbon-11 methionine, for their ability to characterize these tumours for pathways that suggest susceptibility to chemotherapeutic agents not yet studied in osteosarcoma.”
“We want to determine if nonstandard analyses requiring high computational capacities will reveal additional information about these tumours and their response to therapy,” Shulkin said. “In addition, we are planning to evaluate other radiotracers, principally carbon-11 methionine, for their ability to characterize these tumours for pathways that suggest susceptibility to chemotherapeutic agents not yet studied in osteosarcoma.”
- This article was originally published on AuntMinnie.com.
© 2018 by AuntMinnie.com. Any copying, republication or redistribution of AuntMinnie.com content is expressly prohibited without the prior written consent of AuntMinnie.com.