The introduction of PET scans that can image more than one radiotracer simultaneously could revolutionize oncology by enabling the first truly biologically individualized radiotherapy, explains Ceothán Gormley
While modern radiotherapy techniques provide high-precision cancer treatment, cure rates for some advanced cancers have plateaued, with five-year local control rates often remaining as low as 50–60%. Recently, researchers have hypothesized that this clinical resistance may be primarily driven by tumour heterogeneity.
Positron emission tomography (PET) is the gold standard imaging technique for non-invasively mapping biological processes in the body – and could help define tumour regions that may be more resistant to the effects of radiation. Yet conventional scanners remain “monochromatic”, limited to imaging a single radiotracer per session. This physical limitation means that radiotherapy plans are often based on a “one-size-fits-all” dose model that assumes uniform radioresistance across the entire tumour volume.
Multiplexed PET (mPET) is an emerging innovation that offers a significant enhancement by utilizing radiotracers that emit both positrons and gamma photons to detect multiple biological signals simultaneously. The technique holds promise for enabling biologically individualized radiotherapy, allowing for more personalized treatment plans tailored to the unique needs of each patient’s tumour.
Principles of PET
Positron emission tomography (PET) is a widely used functional imaging technique that enables the visualization of metabolic processes within the body. PET imaging relies on electron–positron annihilation, in which gamma-ray photons are emitted when a radiotracer (a pharmaceutical tagged with a positron-emitting isotope, most commonly 18F-fluorodeoxyglucose (18F-FDG)) administered to the patient undergoes beta decay and emits a positron from its nucleus.
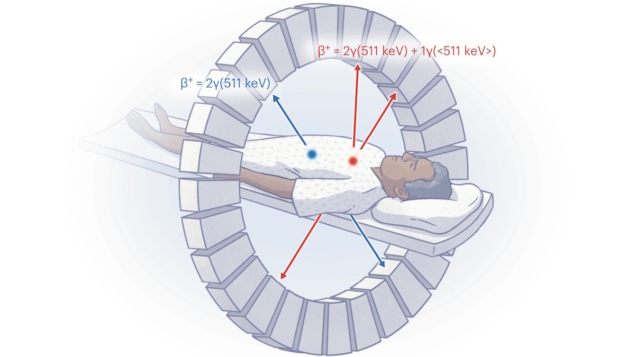
This energetic positron travels a short distance (typically less than 1 mm) through tissue until it encounters an electron in the body. Upon collision, the positron and electron annihilate, converting their mass into energy and releasing two 511 keV gamma photons, emitted approximately 180° apart to conserve momentum. These gamma photons are detected by scintillation crystals in the PET scanner, which convert the photon energy into light. This light is then captured by photomultiplier tubes (PMTs) or silicon avalanche photodiodes (Si-APDs) for precise photon event detection.
The fundamental detection mechanism in PET is coincidence detection, which relies on the arrival of the two photons at opposite sides of the detector ring within a very short time window (typically 6-12 ns). Each coincidence event defines a line-of-response (LOR), which connects the two specific points where the photons strike the detectors. By recording these coincidence events from multiple angles, the system reconstructs a detailed image of the radiotracer’s distribution within the body, allowing for the visualization of physiological processes.
Although PET provides excellent sensitivity for visualizing metabolic activity, conventional single-tracer PET is limited to only one biological process per scan. Since all positron-emitting isotopes produce identical 511 keV photons, standard scanners cannot differentiate between multiple radiotracers based on energy alone. This presents a significant challenge for modern clinical oncology, where tumours exhibit inherent heterogeneity. Different regions within a single tumour often have markedly distinct characteristics, such as variations in oxygenation and vascularization (the network of blood vessels developed by a tumour), which directly influence their radiosensitivity.
For example, hypoxic regions (which lack oxygen) within tumours can increase radiation resistance by up to threefold. While a single radiotracer like FDG can identify metabolically active regions, it does not capture hypoxic, radioresistant areas or variations in clonogenic cell density. This limitation forces radiotherapy to rely on a uniform approach, which often fails to address the complexities of tumour biology.
Sequential imaging with different radiotracers provides more insight into tumour biology but is clinically suboptimal, due to increased radiation burden from multiple accompanying CT scans (used for anatomical registration with the PET images) and higher costs. A method to simultaneously track multiple biological processes in a single scan is needed to fully capture the dynamic nature of tumour biology.
The physical principles of multiplexed PET
In standard PET scans, photons produced by positron–electron annihilation are detected when they arrive simultaneously at opposite sides of a detector ring, defining the LOR. Multiplexed PET builds upon these principles. With dual-tracer PET, however, the detection process becomes more complex due to the need to separate the photon signals from different radiotracers.
To achieve this separation, mPET exploits positron-gamma emitters such as 124I, for instance, which in addition to emitting positrons, emit an additional prompt gamma photon following the positron decay. Such isotopes decay to an excited state of the daughter nucleus, followed by near-instantaneous emission of a de-excitation gamma photon. This additional photon enables the detection of triple coincidence events, providing more biological information in a single scan.
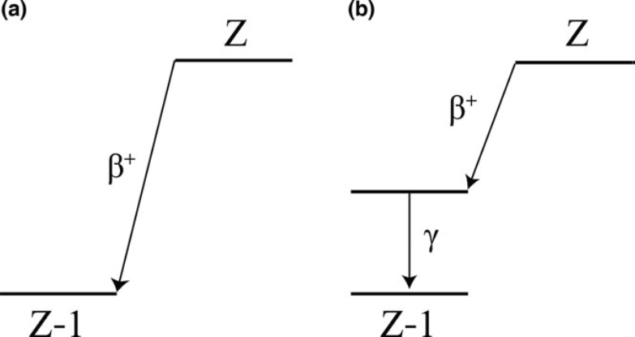
Using a triple-emitting radiotracer in combination with a pure positron emitter enables mPET scanners to achieve effective signal separation by utilizing an expanded energy window (350–700 keV, for example), which enables capture of both the 511 keV annihilation pairs and the higher-energy prompt gamma photons.
These data are then sorted into two streams: the primary dataset, which includes all detected LORs from both isotopes, and a smaller, tagged dataset containing only the triple coincidences. These triple events are identified via a specific timing selection rule, ensuring that the time difference between the prompt gamma detection and the average detection time of the annihilation photons falls within a narrow coincidence window, typically around 4.5 ns.
To reconstruct the separate radiotracer activity distributions, specialized image reconstruction strategies can be used to address the noise and artefacts inherent in basic subtraction methods. One approach is LOR sorting, which compares line integrals from the initial reconstruction to determine the likelihood that a specific LOR corresponds to one of the two isotopes. Furthermore, triple events can be reconstructed using V-shaped LORs, combining two probable LORs from a triple event into a single geometric unit to more accurately approximate the radioactive origin.
This process requires a spatially variant normalization factor that corrects for the camera’s varying efficiency in detecting prompt gammas across the field-of-view, as certain areas may be shadowed by the scanner geometry. Accurate reconstruction must also account for single-photon attenuation correction for the prompt gamma as it travels through the body.
By generating distinct datasets within a single scan, this method provides perfectly co-registered functional maps, allowing clinicians to simultaneously characterize multiple biological processes within a tumour in a single imaging session.
Towards personalized radiotherapy
The introduction of mPET facilitates the transition towards biologically individualized radiotherapy, by delivering perfectly co-registered functional maps in a single imaging session. One promising application is the treatment of head-and-neck squamous cell carcinoma, where the radiotracers 18F-FDG and 18F-FMISO have been used to map clonogenic cell density and hypoxia-related radioresistance, respectively.
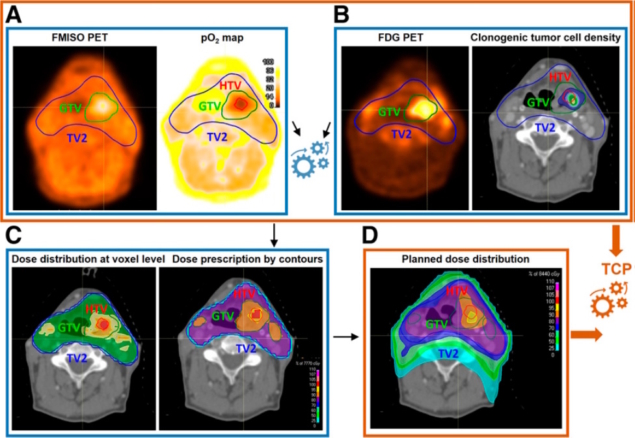
Using radiobiological modelling, radiotracer uptake is converted into voxel-level cellularity maps via linear functions and oxygen partial pressure (pO2) maps via nonlinear sigmoid functions. These biomarkers inform “dose-painting” strategies that strategically escalate radiation to radioresistant areas, such as the hypoxic target volume, while maintaining safe limits for adjacent organs-at-risk. Modelling indicates this synergistic approach could increase tumour control probability from the clinical standard of 60% to a projected 90% or higher.
Researchers have also validated the feasibility of mPET in melanoma mouse models. Here, mPET successfully separated the signals of the triple-emitter 124I-trametinib (targeting proliferation) and 18F-FDG (targeting metabolism). This preclinical trial confirmed that mPET’s ability to separate dual isotopes offers a more detailed and timely assessment of tumour biology.
Future outlook
The clinical translation of mPET represents a significant potential advancement over traditional sequential PET scanning, providing an inherently quicker, cheaper and safer approach. By acquiring dual functional maps simultaneously, the second CT scan required in sequential procedures is no longer needed, roughly halving the patient’s cumulative radiation exposure.
Furthermore, mPET offers the advantage of shorter study duration, as both radiotracers are imaged simultaneously, eliminating the need to wait for the first to decay or wash out before injecting the second. This operational efficiency not only enhances patient compliance but also reduces total costs by minimizing scanner time and overheads. Crucially, mPET is highly viable for near-term implementation as it requires no modifications to existing hardware or acquisition software, when using standard clinical systems, such as the Siemens Biograph mCT, for example.
Despite these advantages, the primary technical pitfall remains the low statistics of the tagged “triples” dataset, which typically represents only a small fraction of total events. This statistical scarcity can introduce significant noise and “shadow” crosstalk artefacts into reconstructed images, potentially affecting quantitative accuracy. To mitigate this, ongoing research into bilateral guided filters and specialized V-shaped LOR algorithms is essential.
In addition, while the physics is compatible with current hardware, many clinical software packages still lack built-in capability for simultaneous multi-energy window acquisition or automated triple-coincidence tagging. This requires the development of manual workarounds that must be standardized for hospital use.
Towards quantum PET: harnessing the diagnostic power of positronium imaging
In the next five to 10 years, as the field moves from discovery into prospective interventional trials, the integration of machine learning for multi-parametric analysis will likely refine signal separation and tumour characterization. Looking further ahead, simultaneous imaging is not necessarily limited to two radiotracers: by utilizing multiple positron–gamma emitters and detecting their unique prompt gamma energies, mPET could evolve into “several-colour” imaging, capable or tracking three or more biological processes at once.
Ultimately, if upcoming trials confirm that predicted gains in tumour control probability translate into actual long-term patient survival, mPET may revolutionize oncology by enabling the first truly biologically individualized radiotherapy.




