A carbon-negative process that produces industrial-scale quantities of acetone and isopropanol has been developed by researchers in the US. Their work could lead to the replacement of some emissions-heavy chemical production methods with more flexible and environmentally friendly processes.
According to the International Energy Agency, the chemical-production sector is the single largest industrial consumer of oil and gas and the third largest carbon emitter, putting out just short of a gigatonne of carbon dioxide emissions in 2020. Acetone and isopropanol (IPA) are two widely employed commodity chemicals used as industrial solvents and platform chemicals for materials production, with a combined global market worth $10bn. Unfortunately, they are manufactured from fossil resources using highly energy-intensive techniques that release hazardous waste and greenhouse gas (GHG) emissions.
As the climate crisis intensifies, researchers are seeking alternative means to lessen the climate impact of producing these commodities. A newly released paper in Nature Biotechnology offers the first report of high-production-rate, high-selectivity, and industrially scalable production of acetone and IPA through fermentation.
Production by fermentation
Acetone and IPA are normally produced by propene cracking and reforming, which are energy-intensive processes with no green alternatives. Scientists have developed methods for biomanufacturing substances by the fermentation of sugars. This involves using natural enzymes and micro-organisms to metabolize these sugars and output the desired products. However, the techniques developed to date have low yields and selectivity, making commercialization unfeasible. Instead, the new study considered autotrophic production, using waste resources such as carbon oxides (carbon monoxide or carbon dioxide) from heavy industry, or syngas from biomass resources, as a starting point.
Co-led by Ching Leang, Michael Jewett and Michael Köpke, the collaborative research team from LanzaTech Inc., Northwestern University, and Oak Ridge National Laboratory achieved industrially relevant productivities of up to about 3 g/l/h with continuous production over about three weeks and high selectivities of up to 90%. These results stand in stark contrast with previous leading rates of 3.8 mg/l/h and 1.2% selectivity.
If at first you don’t succeed; try, try again
In order to achieve such high efficiencies, the team focused its efforts on optimizing the fermentation pathway, microbe strain and process.
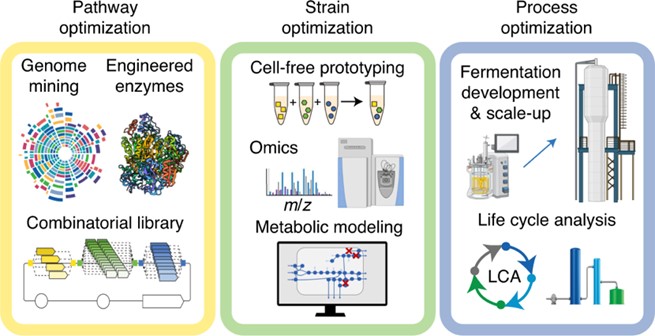
First, the team identified the best set of pathway enzymes to carry out the fermentation, exploring a collection of nearly three hundred biosynthetic genes for acetone production and subsequently screening a library of C. autoethanogenum sAdh enzymes to convert acetone to IPA.
Researchers then optimized the chosen strain to improve selectivity in production – that is to eliminate unwanted byproducts. To edit the strain, they identified and included several genetic modifications, including four gene knockouts and two gene over expressions. To date, previous studies have included a maximum of one genome modification. Including these modifications helped the team increase the proportion of energy in the feed gas that went to the product of interest.
Last, the team transitioned from a two-litre benchtop reactor to a loop reactor in a 120-litre plant. This step helped to demonstrate the scalability of the process and identify problems that might arise when moving from benchtop to industrial production. In particular, bioreactors of this size have spatial heterogeneities — that is, different zones of the reactor have varying chemical compositions. The researchers needed to evaluate the metabolic robustness of the process, analysing how the microbes maintained functionality despite changes in gas substrate concentration. Their observations confirmed that the developed strains could function on an industrial scale.
Greater flexibility
Traditional chemical manufacturing plants are built for single processes. In contrast, bioreactors such as this can be adapted to multiple purposes, offering beneficial flexibility, especially during economic crises when the availability of certain commodities (such as ethanol and IPA in 2019) fluctuate.
Artificial photosynthesis and bacteria transform CO2 into speciality chemicals
Further, gas fermentation is a promising approach from an environmental point of view. While traditional manufacturing of acetone and IPA emits 2.55 kg and 1.85 kg carbon dioxide respectively per kilogram produced, gas fermentation instead is a carbon-negative process as it utilizes waste carbon products. Life cycle analyses conducted by the team showed a negative carbon value; each kilogram of produced acetone and IPA absorbed 1.78 kg and 1.17 kg of carbon dioxide, respectively.
The researchers write that this contrast sparks hope for replacing an emissions-heavy process with a circular economic model “in which the carbon from agriculture, industrial and societal waste streams is recycled into a chemical synthesis value chain and displaces manufacture of products from fresh fossil resources”.