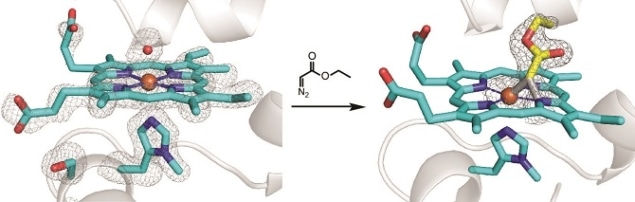
Researchers have successfully engineered a metalloenzyme to extend its natural catalytic functions beyond conventional biological constraints. One crucial aspect of this engineered enzyme is its ability to operate in the presence of oxygen and in the absence of a reductant. The findings by Donald Hilvert’s research group at ETH Zurich lay the foundations for future explorations into biocatalysis, as applied to non-biological chemical reactions that dominate modern chemistry, such as the production of polystyrene and antibiotics.
Enzymes fuelling the future
Metalloenzymes are naturally occurring biological catalysts that have a metal ion incorporated into their structure. This ion helps facilitate a wide range of chemical transformations that are difficult to achieve solely through organic chemistry, which deals with carbon-based compounds. The authors used an expansion of the genetic code to introduce an amino acid that doesn’t normally occur in natural proteins, to create their enzyme. Through genetic coding they were able to fine tune the electronic and bonding properties of the molecule, extending nature’s set of genetically encoded metalloenzymes and opening the door to industrially tailored applications.
Hilvert and his team were also able to glean unprecedented insights into the reaction mechanism studied herein, called styrene cyclopropanation. This has far-reaching implications for catalysts of the future, as enzymes are highly efficient but operate under very specific environments. Better understanding of the properties and reaction pathways that enzymes travel through will lead to better control and applications to a wider variety of chemical processes.

“The most important result of our work is the demonstration that an amino acid can extend the catalytic properties of a metalloprotein in useful ways,” lead researcher Donald Hilvert explains to Physics World.
Hilvert pays homage to other researchers in the field stating that, “extension of these investigations to abiological carbene-transfer reactions was inspired by impressive recent work from Frances Arnold, Rudy Fasan and John Hartwig.”
Reaction routes
Control over the metalloenzyme’s physical properties also opened up a much wider possible operating environment. This makes the molecule oxygen-tolerant, a highly desirable trait for use in industry, as typically the performance of metalloenzymes breaks down in the presence of oxygen.
Genetic manipulation unlocks key to nature’s palette
When investigating the chemical reaction mechanism, researchers were surprised to capture an unusual reaction intermediate consisting of an iron(III) ion bonded with a carbenoid molecule. While this finding goes against previously established beliefs in the field, the researchers confirmed the result using both crystallographic probing and quantum chemical modelling. These investigations point to unexplored potential future avenues for future engineering of catalytic reactions.
In depth studies using an arsenal of techniques comprising quantum calculations and physical characterization enabled the formation of a complete picture of the reaction mechanism, so that researchers could map what is known as a free energy landscape. This tells the story of the reaction in full with each molecular step and transformation accounted for.
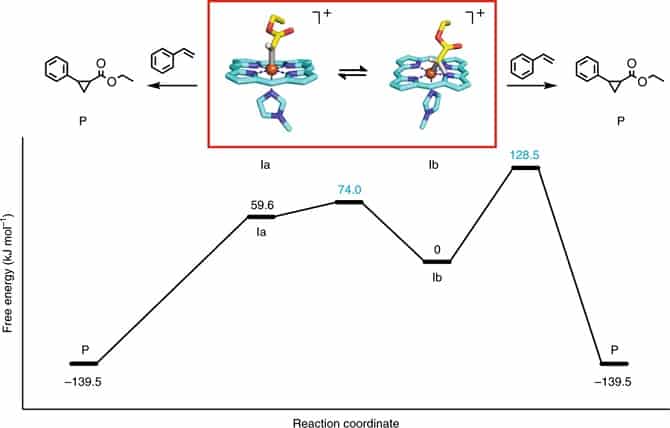
This profound study highlights how genetic manipulation can be used both to engineer high-performance catalysts, and capture and probe unusual reactive species. Deep insights have been gained into biocatalytic processes that will be used in the future. When asked what is next, Hilvert mused that “the ability to tune the reactivity of metal centres with amino acids can be expected to facilitate the creation of novel metalloenzymes for a wide range of synthetically useful biological and abiological transformations.” The future of catalysis is on our doorstep.
Full details of the research are reported in Nature Catalysis DOI: https://doi.org/10.1038/s41929-018-0105-6.