Available to watch now, The Electrochemical Society in partnership with Element6, BioLogic, Gamry Instruments and TA Instruments – Waters, explore delivering technologies that can reduce carbon impact
Want to learn more on this subject?
Whilst many technologies seek to deliver net zero, most fall short when whole life cycle is considered. We therefore need to deliver technologies that actually reduce carbon impact to have a chance of achieving net zero.
In this webinar we present two approaches that can be viewed as illustrative of the negative carbon concept. One is CO2 electrolysis and one is solar conversion of biomass to fuels.
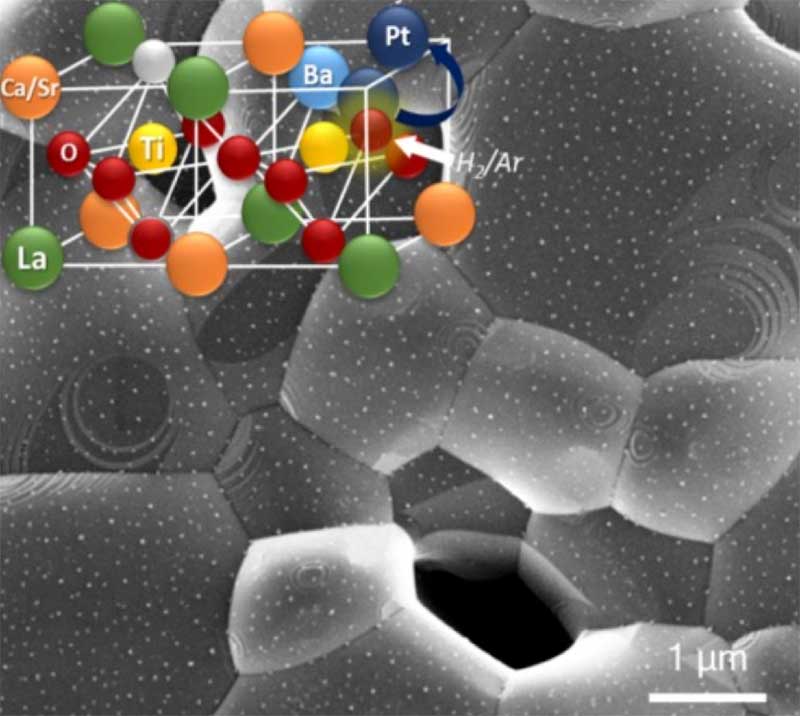
Exsolution under chemical and electrochemical control has been utilised to modify and control solid Oxide electrolysis. Here we utilise this approach to deliver high performance for direct CO2 and steam electrolysis at titanate-based fuel electrodes. Understanding and controlling the processes occurring at electrode/electrolyte interface are key factors in optimising electrochemical conversion devices such as electrolysers. In a new approach, metal particles are grown directly from the oxide support though in situ redox exsolution. We demonstrate that by understanding and manipulating the surface chemistry of an oxide support with adequately designed bulk (non)stoichiometry, one can control the size, distribution and surface coverage of produced particles and so achieve efficient carbon conversions.
Photocatalysis is a versatile technology that has demonstrated potential for solar-driven processes such as water splitting or solar fuels production, and has also been applied to the degradation of pollutants in air and water, and for the production of useful products from biomass. We focus on the products that are produced from cellulose photocatalysis that compliment hydrogen production. We find that an array of oligosaccharides containing only five carbon units initially, with six carbon oligosaccharides later growing to dominate. The photocatalytic process is generally not viewed as a controllable synthetic process; however, these findings show, on the contrary that photocatalysis at semiconductor surfaces can achieve novel reaction pathways yielding new products.
Want to learn more on this subject?

John Irvine FRSE, FRSC has made a unique and world-leading contribution to the science of energy materials, especially fuel cell and energy conversion technologies. This research has ranged from detailed fundamental to strategic and applied science and has had major impact across academia, industry and government. Irvine’s science is highly interdisciplinary extending from chemistry and materials through physics, bioenergy, geoscience, engineering, economics and policy.
The quality and impact of Irvine’s research has been recognized by a number of national and international awards, including the Royal Society Hughes Medal in 2021, the Royal Society of Edinburgh Lord Kelvin Medal in 2018, the Schönbeim gold medal from the European Fuel Cell Forum in 2016, the RSC Sustainable Energy Award in 2015, with earlier RSC recognition via Materials Chemistry, Bacon and Beilby awards/medals.
Highlights of Irvine’s activities include discovery of the emergent nanomaterials phenomenon, establishing the field of oxide fuel electrodes, delivering high-performance direct carbon fuel cells and demonstration of significant hydride ion conductivity. Other important achievements relate to photocatalysis, lithium-ion batteries, non-stoichiometric oxides, structure/property/function, catalysis and electrocatalysis and bioenergy.