Glioblastomas are the most common and deadliest tumours of the central nervous system. Standard-of-care for these tumours typically involves some combination of surgery, radiation therapy and chemotherapy, but patients still often survive only a few months following treatment. Some newer immunotherapy drugs that show promise in other cancers have shown little to no benefit for patients with glioblastomas.
In part, this is because immunotherapies, which boost a person’s immune system so that it can fight cancer, do not cross the blood–brain barrier well. Another challenge is that the tumour microenvironments of glioblastomas suppress the immune system, making it challenging for the immune system to recognize and attack cancerous cells.
A recent study published in ACS Nano presents a novel combination therapy, investigated in mice, that may address both challenges. The therapy, when combined with a burst of radiation, halted glioblastoma growth and prolonged mouse survival.
“We overcame these hurdles by using extracellular vesicles,” says Bakhos Tannous from Harvard Medical School. Tannous, who is senior author on the ACS Nano paper, says that extracellular vesicles (EVs) are “known to facilitate intercellular communications governing diverse processes, such as immune response”.
Therapeutic EVs
EVs are naturally released from many cell types and carry different types of cargo, such as proteins, nucleic acids, lipids and metabolites, from a parent cell. At tens of nanometres to almost 10 µm in size, the smallest EVs can cross the blood–brain barrier and aren’t recognized as invaders.
“They [EVs] are naturally secreted by every cell in the body and therefore are not foreign molecules that induce immune rejection such as solid lipid nanoparticles, for instance,” says Tannous, who is also director of the Experimental Therapeutics Unit and the Viral Vector Core Facility at Massachusetts General Hospital.
Therapeutic uses for EVs arrived on the scene when researchers realized that EVs taken up in a target cell can alter its behaviour. Since this discovery, researchers have demonstrated that EVs can be used as a vehicle to deliver drugs throughout the body.
But EV-based therapies alone are not enough to treat glioblastoma, Tannous’ team notes.
An unwanted brake
Radiation therapy is perhaps the most important nonsurgical treatment for glioblastoma. While radiation sensitizes tumours that fail to generate T cell responses (which help kill cancerous cells), not all responses to radiation are beneficial. Sometimes, infiltrating immune cells are recruited into a tumour in response to radiation. These cells increase the amount of a critical protein called PD-L1.
PD-L1 is often described as a brake that keeps the body’s immune responses under control. Elevated expression of PD-L1 can trick the body’s immune system into thinking that a cancerous tumour isn’t a harmful, foreign substance. As a result, therapy can be less successful than it might be in the absence of elevated levels of PD-L1.
Tannous’ team has introduced a combination immunotherapy that inhibits PD-L1 and induces an immune response in the body to kill cancer cells. Tannous and his collaborators, also based at Nanjing Medical University, the University of Balamand and Brigham and Women’s Hospital, introduced small interfering ribonucleic acids (siRNAs) into EVs to reverse the anti-immunogenic effect that occurs when tumours are primed with radiation.
“By loading EVs with siRNAs against PD-L1 and injecting them in tumour-bearing mice primed with a burst of radiation, we can reverse this effect and induce T cell activation and anti-tumour immunity,” Tannous explains. “The burst of radiation was essential not only in recruiting immune cells, but also in increasing the uptake of these EVs by the tumour and its microenvironment.”
New combination therapy
The team produced the EVs using a human neural progenitor cell line and modified them with a peptide (cyclic RGDyK) that targets brain tumours and helps the EVs penetrate the blood–tumour barrier. The researchers then introduced siRNAs into the EVs’ membranes to help ensure that the body’s immune system responds to the tumour.
The researchers then put their combination therapy to the test. They injected mice with murine glioblastoma cell lines. Murine tumours were primed with a 5 Gy burst of radiation, analogous to stereotactic radiosurgery in a clinical setting, seven and 14 days following tumour cell injection. The mice received an injection of the combination therapy, unmodified EVs or saline (as a control) on days 10, 12, 17 and 19 following tumour cell injection.
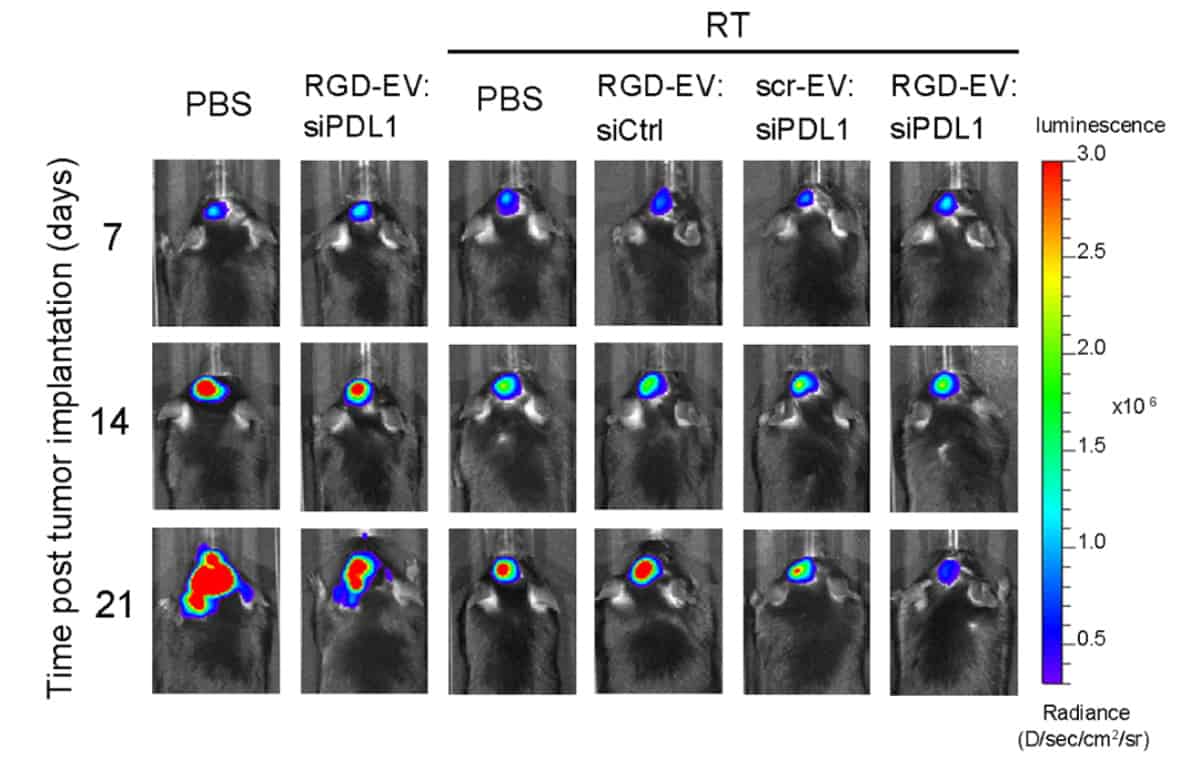
The team found that priming the murine glioblastoma tumours with a burst of radiation enhanced the delivery of EVs to the tumours. The combination therapy also halted tumour growth and prolonged mouse survival. Results further suggest that using EVs allowed the immunotherapy to cross the blood–brain/tumour barrier, recruiting immune cells to the tumour site and inhibiting the expression of PD-L1. Fluorescence imaging studies showed that stronger fluorescence signals, and therefore more EVs, were observed in mice brains that had been irradiated relative to those that were not.
Large-scale production
The researchers say that their decision to isolate EVs from a human neural progenitor cell line, rather than stem cells or dendritic cells, was an important one. Using a human neural progenitor cell line allows them to produce EVs in larger quantities for larger studies and clinical trials.
LATTICE radiotherapy plus immunotherapy shows promise for treating advanced bulky tumours
Another important decision was to use copper-free click chemistry to modify EVs to include the brain-tumour-targeting peptide on the EV surface. Copper-free click chemistry, unlike some cell engineering methods, is suitable for in vivo applications, is fast and can be used for large-scale production of modified EVs. Previous work from the group found no obvious toxicity or tissue damage using these methods.
Now, Tannous’ team is scaling up EV production and labelling. The researchers are also working to further improve the EV delivery system and are testing how EVs can deliver nucleic acid therapies to brain tumours.