Conventional medication is often limited by low drug effectiveness or intolerable side effects caused by the drug reaching parts of the body where it’s not needed. As such, there’s increasing interest in developing methods for targeted drug delivery, to increase the efficacy and safety of pharmacological treatments.
One promising approach for localized drug delivery lies in the use of low-intensity ultrasound as a safe and practical way to trigger targeted drug release from circulating nanocarriers. Previous investigations using perfluorocarbon (PFC) nanodroplets as the drug carriers, however, have proved either effective or safe, but not both.
In their latest study, researchers from the University of Utah have shown that such PFC nanodroplets can be activated using low-frequency ultrasound, providing effective drug release with a favourable safety profile. Writing in Frontiers in Molecular Biosciences, the team also describes an optimized method for reliable manufacture of the nanocarriers, to help in the clinical translation of this approach.
“Delivery of medication into specific parts of the body has been a dream of medicine,” says Jan Kubanek, the study’s senior author. “This prevents the problems associated with taking drugs in the common way, which affects the entire body and all organs, often triggering substantial side effects.”
Drug release mechanisms
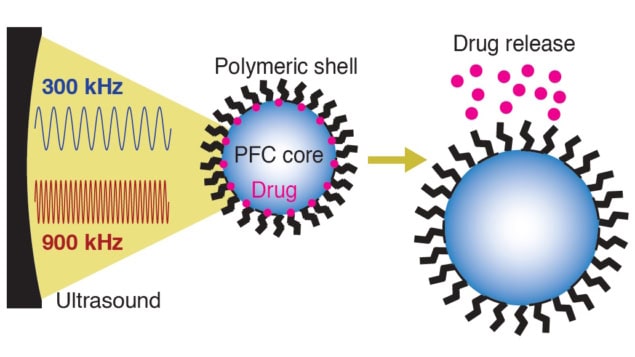
The nanocarriers comprise polymeric nanodroplet shells, of roughly 500 nm in diameter, filled with inert PFC cores mixed with a drug. When exposed to ultrasound, the PFC cores expand, stretching the droplet’s shell and releasing the encapsulated drug. This expansion arises due to either the mechanical or the thermal effects of ultrasound, with high-frequency ultrasound accentuating thermal mechanisms and low-frequencies causing mechanical disruption.
Most studies to date have investigated nanodroplets with PFC boiling points below body temperature, activated by high-frequency (1 MHz or greater) ultrasound. These low-boiling point PFCs, however, run the risk of spontaneous drug release – having been observed to more than double in size after 24 h at room temperature. High-boiling point PFCs are safer and more stable but to date have not demonstrated effective drug release, likely because they are less affected by the thermal mechanisms associated with high-frequency ultrasound.
To identify suitable ultrasound parameters to trigger drug release from high-boiling point nanodroplets, the team examined three different PFC cores – perfluoropentane (PFP, with a boiling point of 29°C), decafluoropentane (DFP, 55°C) and perfluorooctylbromide (PFOB, 142°C) – loaded with the anaesthetic drug propofol.
The researchers measured the level of drug released into organic solvent upon exposing the nanodroplets to either low-frequency (300 kHz) or high-frequency (900 kHz) ultrasound. At 900 kHz, they observed an increase in drug release with decreasing PFC boiling point, supporting the hypothesis of a thermal mechanism. They also saw a quadratic dependence of drug release on ultrasound pressure, in accordance with the fact that ultrasound-delivered thermal energy is proportional to pressure squared.
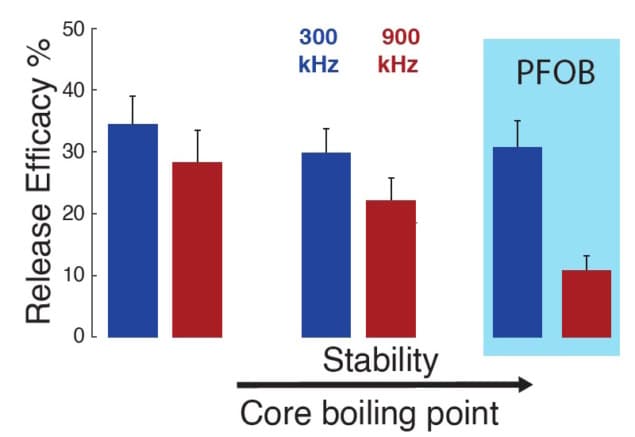
In contrast, sonication at 300 kHz produced a linear dependence of drug release on ultrasound pressure, consistent with a mechanical rather than a thermal effect. The researchers also found that for all three PFC cores, 300 kHz ultrasound released a greater percentage of drug (an average of 31.7%) than 900 kHz (20.3%).
These findings suggest that low-frequency ultrasound could open the path to using stable, high-boiling point cores such as PFOB as release actuators. The mechanical nature of drug release at low frequencies could also be of benefit for clinical translation, as it does not induce any heating of the target tissue.
“We developed a method to produce stable nanocarriers repeatably, and identified ultrasound parameters that can activate them,” says first author Matthew Wilson in a press statement.
Safe and sound
Kubanek and colleagues evaluated the safety of the high-boiling point PFOB-based nanodroplets in the brain of a macaque monkey. They delivered six doses at one-week intervals and analysed blood samples for markers of toxicity to the liver, kidney or spleen, as well as assessing immune system activation.
The blood biomarkers showed that the nanodroplets were well tolerated, with no detectable side-effects, an important factor for potential clinical translation. Only blood glucose showed a significant change following administration, which may have been caused by the animal’s diet.
Bacteria-based microrobots show potential for cancer drug delivery
The team also demonstrated that PFOB nanodroplets loaded with the anaesthetic ketamine or an immunosuppressant drug exhibited a similar sensitivity to ultrasound as propofol-loaded nanodroplets. “We are now testing the release of other psychoactive drugs, including ketamine, in non-human primates,” says Kubanek. “If we could release these locally, we would avoid the commonly observed side effects and thus open these emerging treatments of mental disorders to many more people.”
The researchers have now begun investigating the delivery of anti-cancer drugs into the brains of mice with glioblastoma. Kubanek notes that the nanocarriers could also be used to target drugs to tumours elsewhere in the body. “Through these developments in rodents and non-human primates, we are aiming to bring the method to humans as soon as possible,” he tells Physics World.