Blood flow and oxygen supply to tumours change over the first few weeks of radiotherapy. Scientists currently believe that reoxygenation occurs due to tumour shrinkage, decreased oxygen consumption and increased perfusion. Clinicians hope that these and other possible changes might be exploited to improve a patient’s response to radiation therapy.
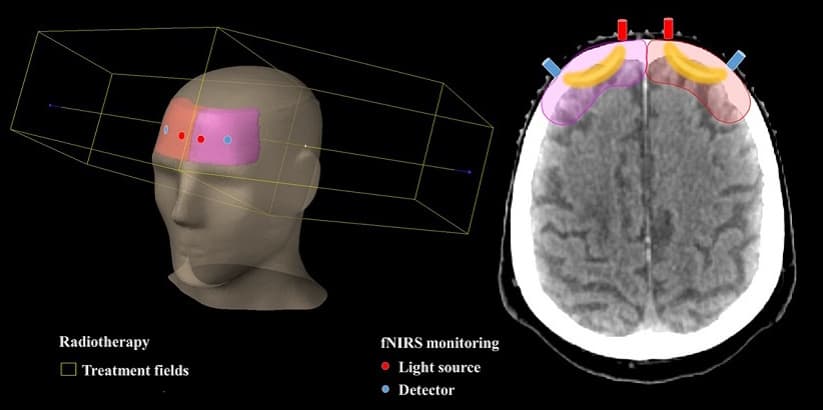
In a potential step toward personalized cancer treatments, researchers in Finland are using functional near-infrared spectroscopy (fNIRS) to measure real-time haemoglobin concentration, an indirect indicator of tissue oxygenation, during whole-brain radiotherapy.
Teemu Myllylä from the University of Oulu leads the fNIRS research in collaboration with Juha Nikkinen, chief physicist in the clinical medical physics division of radiotherapy at Oulu University Hospital. The goal of their research studies, Myllylä says, is to apply fNIRS to begin to close some of the gaps in our knowledge of tissue and tumour oxygenation and response during and after radiotherapy.
Functional near-infrared spectroscopy has been used for several decades to study real-time brain activity in response to different stimuli and cognitive tasks. The relatively inexpensive, portable and non-invasive approach can measure cerebral haemodynamics up to 2 cm deep in the adult human brain. The fNIRS device uses infrared light to measure real-time changes in regional concentrations of haemoglobin – a surrogate for changes in blood volume and, by extension, how well oxygen is getting to tissues – in the brain.
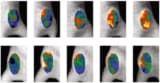
In their recent proof-of-concept study, reported in the Journal of Biomedical Optics, the researchers used fNIRS to measure haemoglobin concentration during palliative whole-brain radiotherapy. The team observed increased blood flow during treatments in 10 patients undergoing multiple whole-brain irradiations. No effect was observed before irradiation or after irradiation ended.
The team attached fibre optic tips for the multiwavelength fNIRS device perpendicular to the brain and confirmed that they did not interfere with the setup or delivery of radiation. Radiation dose was delivered using static field whole-brain radiotherapy, which comprised two opposing 6 MV fields. Forward-intensity-modulated radiation therapy, which adds smaller fields from the same direction as the main fields, was applied to provide homogeneous dose coverage of the entire brain.
Because a NIRS device only measures the relative concentration of haemoglobin throughout the brain, different patients have different fNIRS signal amplitudes. The researchers normalized the signal amplitudes by filtering the fNIRS signal in a very low-frequency band and then subtracting the signal at the beginning of the irradiation from whole corresponding signals. They used resting state data from hundreds of healthy individuals as control data.
Tumour hypoxia tracked in real time during radiotherapy
The team is now collecting fNIRS data from participants with solid tumours to try to differentiate between haemoglobin concentrations in tumour and healthy tissue and to study tumour responses to irradiation. They are also investigating why they observed differences in tissue oxygenation between the first and second irradiation in their Journal of Biomedical Optics study. Possible explanations include a smaller absorbed dose in the second irradiation, multileaf collimator or other measurement setup effects, or physiological responses.
“The [fNIRS] technology is easy to perform in clinical settings and practically does not disturb or slow down the normal radiotherapy procedures performed on patients,” Myllylä says. “There is high potential to exploit fNIRS in clinical situations because it is a safe technique and can be used in combination practically with all currently-used clinical neuroimaging and therapy techniques.”