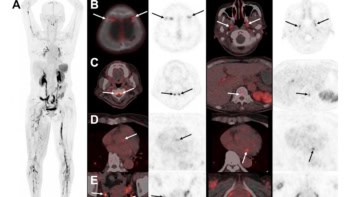
The Annual Meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI) takes place this week in Vancouver, Canada. Each year, the SNMMI chooses an abstract that represents the most promising advances in the field of nuclear medicine and molecular imaging. This year, the Abstract of the Year was awarded to researchers at the All India Institute of Medical Science (AIIMS) in New Delhi, for their study of a targeted radionuclide treatment for advanced neuroendocrine tumours.
Gastroenteropancreatic neuroendocrine tumours (GEP-NETs) are rare malignancies that arise from neuroendocrine cells and can occur anywhere along the gastroinstesinal tract. While surgery can cure early-stage GEP-NETs, most patients are diagnosed with metastatic disease, making systemic treatment such as targeted radionuclide therapy their only option.
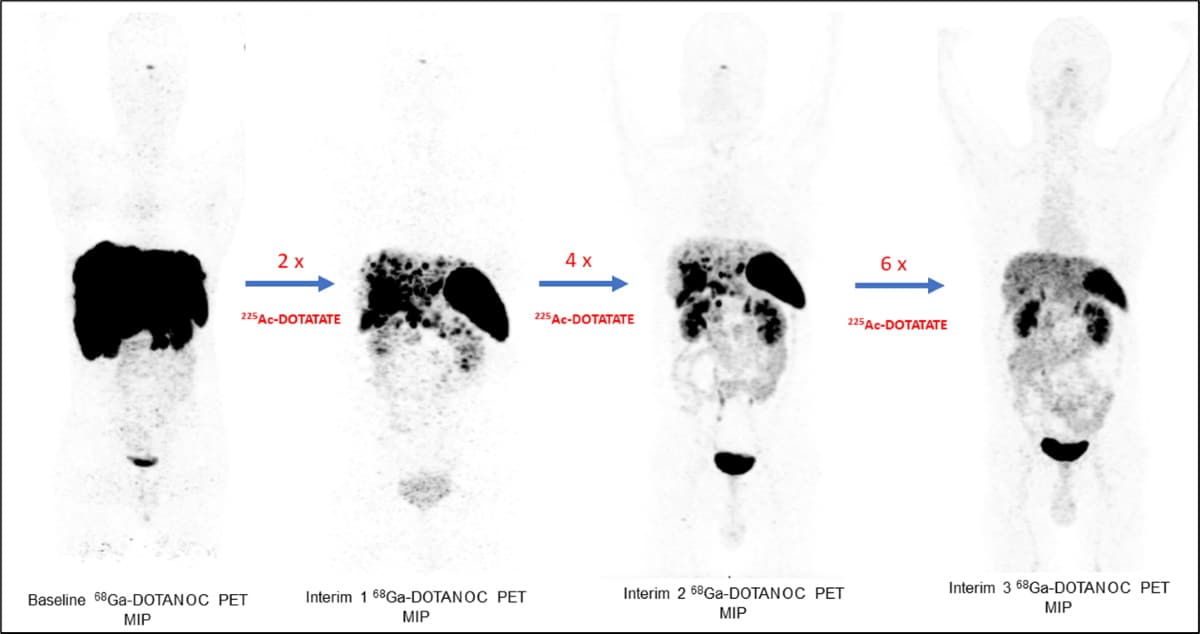
In this Phase II study, the researchers demonstrated that the targeted radionuclide alpha therapy 225Ac-DOTATATE has long-term anti-tumour effects in patients with advanced GEP-NETs. They evaluated the long-term efficacy, survival outcomes and safety of 225Ac-DOTATATE in 83 GEP-NET patients, who received systemic treatment with intravenous 225Ac-DOTATATE at eight weekly intervals. After the treatment course, two patients (2.7%) had complete response, 32 (43.2%) had a partial response, 25 (34%) had stable disease and 15 (20%) had progressive disease. The team notes that there was minimal toxicity from the treatment.
The study results suggest that 225Ac-DOTATATE could provide a potential treatment, even for patients who were resistant to prior therapy with 177Lu-DOTATATE (a beta-emitting radionuclide). “This is a promising therapy option that adds a new dimension to the treatment of end-stage GEP-NETs, especially for patients who have tried all other standard therapy options,” said Chandrasekhar Bal, head of the department of nuclear medicine and PET at AIIMS. “These results warrant a Phase III randomized control trial to assess the true efficacy of 225Ac-DOTATATE versus 177Lu-DOTATATE.”
Targeting prostate cancer with alpha-emitters
In another study of targeted radionuclide alpha therapy, a team from the University of Wisconsin-Madison showed that treatment of advanced prostate cancer with the alpha emitter 225AC-NM600 achieved significantly better outcomes than its corresponding targeted radionuclide beta therapy.
“Metastatic castration-resistance prostate cancer is the most lethal form of the disease and has a median life expectancy of less than five years,” said Carolina Ferreira. “Innovative targeted therapies to treat this advanced form of cancer are needed to significantly improve survival.”
Exploiting the fact that prostate cancer cells selectively sequester and retain alkylphospholipds, Ferreira and colleagues developed an analogue – NM600 – to target prostate cancer cells. They then paired NM600 with alpha-emitting (225Ac) and beta-emitting (177Lu) isotopes to create two types of targeted radionuclide therapies, and compared their effectiveness in two mouse models of prostate cancer.
The researchers treated the mice with varying doses of 225Ac-NM600 or 177Lu-NM600, performing PET/CT scans to image the tumours. Targeted radionuclide therapy with the alpha-emitter produced significantly better outcomes, such as slowed tumour growth and improved overall survival, than treatment with the beta-emitter at similar absorbed doses in both tumour models. Both treatments were well tolerated by the animals.
“This study shows that targeted radionuclide therapies with alpha- and beta-emitters have distinctive, often unexpected, effects on the tumour microenvironment,” noted Ferreira. “Careful exploration of combination regimens, such as targeted radionuclide therapies with anti-tumour vaccines or checkpoint blockade, is warranted.”
PET monitors post-COVID lung disease
Elsewhere at the SNMMI Annual Meeting, researchers from the Sanjay Gandhi Postgraduate Institute of Medical Sciences in India described a study using PET/CT to evaluate and monitor post-COVID-19 lung disease. They showed for the first time that residual COVID-19 symptoms, such as cough and breathlessness, can be attributed to ongoing inflammation in the lungs.

“During the pandemic, there was no standard modality to assess residual lung inflammation. It was difficult to gauge the extent and severity of disease even in recovering patients, and hence challenging to start appropriate treatment,” explained Yogita Khandelwal.
In the study, Khandelwal and colleagues used 18F-FDG PET/CT to assess the metabolic activity of lung lesions and evaluate the effect of treatment with steroids and antifibrotic drugs. They performed baseline scans to evaluate the residual inflammatory activity in the lungs of 25 patients with post-COVID-19 lung disease. Those with inflammation received steroid and antifibrotic treatment, with a follow-up 18F-FDG PET/CT scan six to 12 weeks later to evaluate treatment response.
Targeted treatment guides radiation directly to pancreatic tumours
All patients initially showed metabolically active lesions in both lungs, 13 also had metabolically active mediastinal lymph nodes. After treatment, the team observed a significant decrease in the number, size and FDG-avidity of the lung lesions in the 22 surviving patients.
Khandelwal concluded that 18F-FDG PET/CT provides a sensitive tool for monitoring ongoing inflammation and its management. “Molecular markers identified by 18F-FDG PET/CT can reveal the triggers and sustenance mechanism of inflammation,” she said. “In the future, this could contribute to the development of new drugs and better management strategies.”