Life as we know it would not be possible were it not for a particular nuclear energy level of carbon-12 predicted 60 years ago by Fred Hoyle. But the true nature of this energy level remains one of the biggest unsolved questions in nuclear physics, say David Jenkins and Oliver Kirsebom

Without carbon, life as we know it on Earth would not exist. But carbon itself would not exist in sufficient quantities for life were it not for a strange feature of its nuclear structure, which has yet to be fully understood. This mystery surrounds one particular excited state of the carbon-12 nucleus – a special arrangement of its protons and neutrons – which our basic models say should not even be there, but is. An essential step in carbon creation within the depths of red giant stars, the nature of this excited state is one of the most intensely researched questions in nuclear physics.
Only three elements were produced in the Big Bang – lots and lots of hydrogen and helium, plus a tiny amount of lithium. Since the 1950s we have known that heavier elements, such as carbon, oxygen and silicon, are produced through nuclear fusion in stars. But as the scientific understanding of these fusion processes began to evolve, scientists also wanted to know why and how the elements form in the abundances in which we find them.
We now know that stellar nucleosynthesis begins with four hydrogen nuclei (four protons) fusing together to form a helium nucleus (two protons and two neutrons). Heavier elements are then created when these alpha particles fuse at high temperatures in massive stars, leading to nuclei such as beryllium-8, carbon-12 and oxygen-16, which have twice, three times and four times the number of protons and neutrons as alpha particles. However, there is a problem. The first step in any of these reactions involves two alpha particles fusing to create beryllium-8, but this nucleus is extremely short-lived, with a lifetime of only 8 × 10–17 s. It therefore does not usually hang around long enough for a third alpha particle to fuse together with it to form carbon-12. Indeed, the probability of carbon-12 forming should be tiny and could not account for the observed abundances.
To try to explain this mystery, in 1953 the British astronomer Fred Hoyle reasoned that because we exist and carbon manifestly exists, there must be something that makes the production of carbon in stars proceed at a much faster rate than would appear likely at first glance. To understand what that something is, recall that nuclei – like atoms – have a discrete set of energy levels consisting of a ground state plus many excited states. Hoyle predicted the existence of a very short-lived excited state in carbon-12 that serves as an intermediate step in the synthesis of carbon.
This fleeting “Hoyle state”, which would decay by emitting gamma rays to reach the stable ground state of carbon-12, would serve as a “resonance” that would accelerate production of carbon-12 by seven orders of magnitude. However, for this to work the Hoyle state would need to have certain properties, including a specific excitation energy.
To understand why, recall that some nuclei have larger binding energies than others. In particular, carbon-12 has 7.28 MeV more binding energy than an alpha particle and a beryllium-8 nucleus combined. So when an alpha particle and a beryllium-8 nucleus, both at rest, fuse they form a carbon-12 nucleus with an excitation energy of exactly 7.28 MeV – because energy has to be conserved. In stars, however, nuclei are not at rest but have some thermal energy, which they bring with them when they fuse, resulting in a slightly higher excitation energy. Hoyle calculated that the excitation energy of this short-lived state had to be 7.68 MeV, plus or minus a few per cent. The Hoyle state should also have a nuclear spin of zero and positive parity.
Hoyle badgered his experimental colleague, Ward Whaling from the Kellogg Radiation Laboratory at the California Institute of Technology, to search with his team for such a state, which they did by bombarding a nitrogen-14 target with deuterons (which contain one proton and one neutron). Using a magnetic spectrometer, they then measured the energy spectrum of the alpha particles produced alongside carbon-12, which – using energy conservation – revealed a state at the precise excitation energy suggested by Hoyle.

So had the carbon-production conundrum now been solved? In a way, yes – scientists could now rationalize that, via the Hoyle state, carbon can be formed fast enough to overcome the fact that beryllium-8 is so short-lived, resulting in the abundance of carbon we see around us. However, a new puzzle immediately emerged that, 60 years on, has still not been resolved. Quite simply, the Hoyle state cannot be described by any known models of atomic nuclei. Given how important carbon is for life to form, this situation is deeply unsatisfying, though it is not for want of effort. Indeed, trying to work out the precise nature of the Hoyle state remains one of the most intensive theoretical and experimental efforts in nuclear physics.
Not your normal nucleus
So what picture do we have of nuclei? Our understanding of the nucleus is underpinned by the “nuclear shell model”, which was developed in 1949 – four years before the discovery of the Hoyle state. The model, which was a triumph in our understanding of nuclear structure, says that protons and neutrons act like independent particles that fill up shells rather in the same way that electrons do in atoms, with simple nuclear states constructed from configurations of protons and neutrons in different shells. This model is remarkably successful in describing ground states and excited states, especially in light nuclei. It also does a reasonably good job of describing most of the lowest excited states in carbon-12. The nuclear shell model is, however, hopelessly inaccurate for the Hoyle state. Quite simply, none of the energy levels that the model calculates are remotely close to the energy of the Hoyle state. Indeed, there are similar states in other light nuclei where the shell model also fails badly.
But could it be that the Hoyle state is better described in terms of clusters of alpha particles rather than in terms of nucleons as independent particles? Indeed, in 1956 – only three years after the prediction and discovery of the Hoyle state – Haruhiko Morinaga, who was then at Purdue University, Indiana, conjectured that the Hoyle state could be thought of as a linear chain of three alpha particles. Similar states composed of four, five and six alpha particles, he argued, should also exist in oxygen-16, neon-20 and magnesium-24.
From the beginning, it was clear that the static, linear-chain structure proposed by Morinaga had to be a gross over-simplification. The alpha particles would have to be in continuous motion so as not to violate Heisenberg’s uncertainty principle: since they are confined within a small distance – a nucleus no larger than a few femtometres – their momentum and therefore speed must be above a certain value. The alpha particles might even exchange nucleons between one another. In other words, Morinaga’s alpha-cluster model merely represented the time-averaged picture of a dynamic system.
Our current “best understanding” is that the Hoyle state is a gas-like system of weakly interacting alpha particles that move almost freely over distances that are large on nuclear scales. Moreover, within the past decade, it has become clear that the Hoyle state is likely to exist as a superposition of states: in the same way that Schrödinger’s cat is 50% dead and 50% alive, the Hoyle state is about 70% a state of three alpha particles and 30% a shell-model-type state (in which individual nucleons fill up quantized energy levels).
The only other strong contender for a competing theory is an evolution of the alpha-cluster model in which the Hoyle state could be a Bose–Einstein condensate (BEC). This idea arises because alpha particles have zero spin, which means that they are bosons and obey Bose statistics. A system of alpha particles with a low internal temperature might therefore form a BEC, similar to the atomic condensates that are nowadays routinely made and manipulated in laboratories. This idea has attracted much attention but also faces many difficulties. Nuclear condensates, if they exist, would differ from their well-established atomic counterparts in many respects, and it is not clear how, or if, the theory of atomic condensates can be adapted to the nuclear domain.
Recent revelations
Current-day Hoyle-state physicists are trying to get to the bottom of this mystery once and for all. Theorists dream of being able to predict the properties of nuclei from first principles, which requires two ingredients: a theory of how the protons and neutrons in the nucleus interact and a method to solve the many-body Schrödinger equation numerically. The first ingredient was provided by Steven Weinberg in 1991 in the form of the “chiral effective field theory”. For many years the second ingredient seemed a distant prospect, but the enormous growth in computer power has recently allowed nuclear physicists to make some hugely exciting theoretical simulations.
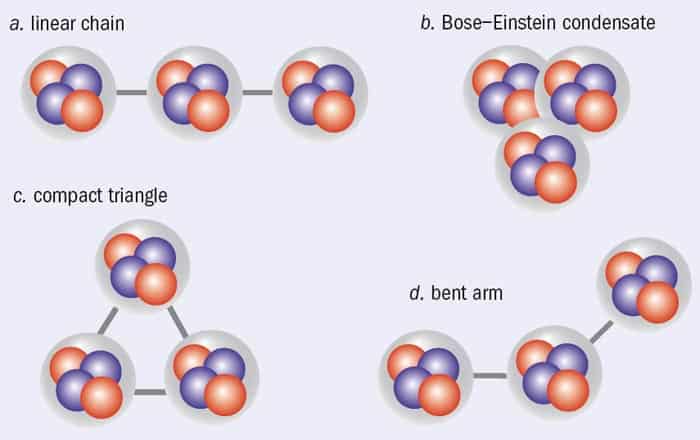
In 2011, for example, a team led by Evgeny Epelbaum, from the Institute of Theoretical Physics II at Ruhr-Universität Bochum in Germany, performed the first calculation of the Hoyle state from first principles, using a computational method known as Lattice Monte Carlo simulation and a nuclear interaction derived from quantum chromodynamics – the theory of quarks and gluons – using Weinberg’s approach. Remarkably, they found a state with zero nuclear spin and positive parity about 7 MeV above the ground state that appears to be the Hoyle state. And only a few months ago, Epelbaum and colleagues published new improved calculations for the Hoyle state (2012 Phys. Rev. Lett. 109 25201) in which the alpha-cluster structure emerges naturally from their calculations rather than having to be “put in by hand” as is usually the case. According to Epelbaum’s work, the alpha particles in the ground state of carbon-12 appear to be arranged in a compact triangle, whereas in the Hoyle state, they are in a bent-arm configuration (figure 1).
As well as advances in theory, there have been important strides in experiment. Unfortunately, as we cannot use a “nuclear microscope” that lets us see directly what is going on inside the Hoyle state, we have to rely on indirect experimental evidence of its structure. Physicists have, for example, determined the excitation energy of the Hoyle state to a precision of 0.05% and they have measured a very rare decay mechanism seen in only seven in one million decays of the Hoyle state, where it relaxes into the carbon-12 ground state by simultaneously emitting an electron and its antimatter counterpart, the positron.
Nevertheless, 60 years on from the verification of the existence of the Hoyle state, experiments are only just beginning to provide discriminating tests of its true nature. By studying its properties in detail, physicists hope to figure out if the alpha-cluster description is the best model for the Hoyle state, and if so whether the alpha particles behave like a gas or a BEC.
One promising line of experimental inquiry relates to rotational excitations of the Hoyle state. This story dates back to 1956, when Morinaga conjectured that the Hoyle state might consist of a linear chain of three alpha particles, and that – just like a chemical molecule – it should therefore be possible for this structure to rotate. If this were to happen, it would lead to a set of quantized energy levels with spin and parities of 2+, 4+, 6+, etc. The spacing of these levels is related to the moment of inertia associated with the Hoyle state, which would tell us something about how compactly the alpha particles are arranged.
This was a tangible prediction and experimentalists quickly began looking for these “rotational excitations” of the Hoyle state. Sadly, more than half a century later the search is still on – showing just how hard the task is and the importance of the result in pinning down the structure of the Hoyle state. But efforts have not been abandoned; most people think the states exist but are just incredibly difficult to identify experimentally. In fact, the search is being conducted more vigorously now than ever before, with as many as 10 experiments performed in the past decade. An important recent result has been a candidate 2+ state identified by Martin Freer of the University of Birmingham in the UK and his team in an experiment conducted at the iThemba Laboratory for Accelerator-Based Sciences in South Africa.
A second strand of experimental endeavour has been to study the break-up of the Hoyle state into its constituent alpha particles. Recently, one of us (OK) led an experiment to scrutinize the break-up mechanism. It was already known that the Hoyle state preferentially decays to the ground state of beryllium-8, which subsequently breaks up into two alpha particles. We wanted to see if, on rare occasions, the decay to three alpha particles would proceed differently, bypassing the ground state of beryllium-8. We found that, at least 99.5% of the time, the Hoyle state decays via the ground state of beryllium-8. If an experiment with higher sensitivity could be designed, it might be possible to discover other, very rare, decay mechanisms. This could shed light on the current controversy regarding the nature of the Hoyle state – BEC or “ordinary” alpha-particle gas?
Understanding the origin and structure of the Hoyle state continues to pose a strong challenge to theory and attracts considerable experimental efforts. At last year’s international Cluster12 nuclear-physics conference, for instance, discussion of the nature of the Hoyle state was a major focus of the meeting and dominated the first day. Curiosity still abounds 60 years on from Fred Hoyle’s prediction of his eponymous nuclear state, as we strive to finally understand how carbon, and thus life, came to be.
Anthropic musings
Were it not for a particular excited state of the carbon-12 nucleus known as the Hoyle state, the universe would look very different. In particular, there would be essentially no carbon and hence life as we know it would not have evolved, nor would other heavy elements exist. In hindsight, it is remarkable that the laws of nature have conspired to create an excited state in carbon-12 that allows carbon to be produced in significant quantities in stars.
Some people have discussed this situation in terms of the anthropic principle. This is the point of view that the physical parameters of the observed universe must be compatible with our existence as conscious observers; if the physical parameters had been different in a way that prevented life from forming, we simply would not be around in order to measure them.
The situation becomes more remarkable still when one considers that the rate at which three alpha particles fuse into carbon is incredibly sensitive to the energy of the Hoyle state. The Hoyle state is 7.65 MeV above the ground state of carbon-12, but if it were a mere 0.06 MeV more or less, the abundance of carbon in our universe would be very different.
In 2010 Sylvia Ekström, an astrophysicist at the Observatory of Geneva, and colleagues used this property of the Hoyle state to test whether the strengths of electromagnetic and nuclear interactions might vary in time and space, rather than be constant as assumed. Ekström and her team observed abundances of carbon and oxygen on the surfaces of very old stars in the galactic halo, which were formed from interstellar dust only 200–300 million years after the Big Bang. This allowed them to determine by how much the energy of the Hoyle state could have been different back then. Since the energy of the Hoyle state ultimately depends on the interplay between the electromagnetic and nuclear interactions, they were able to relate this to a change in the interaction strengths. Their conclusion: there is no evidence that the strengths of the electromagnetic and nuclear interactions were any different in the early universe, and the most they can have changed by is 0.001% and 0.1%, respectively.