Pufferfish have inspired a new device that is capable of the highest rate ever of passive solar water purification – according to its creators Xiaohui Xu and colleagues at Princeton University in the US. Based on an advanced hydrogel, the system can rapidly soak up and filter water when cool, and then release clean water when warmed in the Sun. The team hopes their innovation could lead to low-cost and sustainable off-grid purification systems, potentially improving access to clean water for many communities worldwide.
According to the United Nations, one in three people worldwide do not have access to safe drinking water. Therefore, there is a pressing need for inexpensive, environmentally sustainable systems for filtering water. These are best achieved through passive purification technologies, which use solar energy to separate water from contaminants such as heavy metals, oils, and harmful microbes. Today, this is widely done by evaporating water, and condensing it onto a surface – but this is an energy intensive process, leading to slow production rates.
Xu’s team has taken a more advanced approach based on the behaviour of pufferfish. When these fish detect predators, they rapidly absorb water to swell their bodies, making themselves appear more threatening. Once danger passes, the water is quickly released. To mimic this behaviour, the researchers developed a sponge-like device they dubbed a solar absorber gel (SAG), containing three key components.
PNIPA polymer chains
At the centre of the material is an advanced hydrogel, composed of a temperature-sensitive mesh of PNIPA polymer chains, containing both hydrophobic and hydrophilic regions. At lower temperatures, these chains remain long and flexible. This enables water to flow into the mesh through capillary action, and bond with its hydrophilic regions. At temperatures higher than 33 °C, the PNIPA chains undergo a phase transition, becoming short and rigid. As a result, the mesh loses around 90% of its volume and it becomes hydrophobic – pushing water out of the material.
Surrounding this inner hydrogel is a dark layer of polydopamine, which efficiently converts sunlight into heat: enabling the PNIPA to reach its phase transition temperature in cooler conditions, while also filtering out heavy metals and organic molecules before they entered the hydrogel. Finally, an outer alginate layer filtered out any microbes, along with any other larger molecules.
Nanoparticle sensors detect arsenic in drinking water
Xu’s team tested the SAG’s performance by placing it in a lake on the Princeton campus with a water temperature of 25 °C. After soaking the material, they warmed it in sunlight to release its absorbed water. Over a 2 h cycle of soaking and discharge, the material demonstrated the highest rate of passive solar water purification ever reported. The SAG was also highly durable; barely diminishing in performance even after 10 collection cycles.
Owing to their simple, water-based manufacturing process, SAG materials are low-cost and non-toxic, ensuring both their accessibility and sustainability for off-grid water purification. Xu and colleagues believe their material could be transformative: potentially improving quality of life for many communities around the world, especially where access to electricity is limited.
The research is described in Advanced Materials.
 This webinar will address the critical role of independence in providing unbiased assurance in radiation therapy that treatment-related issues are caught. While vendor-integrated system self-checks represent some efficiency gains, independent quality assurance and the data accessibility required for it remain essential – especially as treatment complexity increases.
This webinar will address the critical role of independence in providing unbiased assurance in radiation therapy that treatment-related issues are caught. While vendor-integrated system self-checks represent some efficiency gains, independent quality assurance and the data accessibility required for it remain essential – especially as treatment complexity increases. Jeff Kapatoes received his PhD in medical physics in 2000 from the University of Wisconsin as part of the tomotherapy research team. Upon graduation, he joined start-up TomoTherapy Inc, spending 10 years with the company, culminating with his management of the software development team, which included responsibility for treatment planning, optimization, database services, treatment delivery, imaging and calibration. Kapatoes then worked at Mevion Medical Systems as the director of project management and helped the company secure its initial FDA 510(k) clearance. In 2012, he joined Sun Nuclear Corporation as the product manager of its Patient QA products. He continues to work for Sun Nuclear today as the senior director of research and regulatory.
Jeff Kapatoes received his PhD in medical physics in 2000 from the University of Wisconsin as part of the tomotherapy research team. Upon graduation, he joined start-up TomoTherapy Inc, spending 10 years with the company, culminating with his management of the software development team, which included responsibility for treatment planning, optimization, database services, treatment delivery, imaging and calibration. Kapatoes then worked at Mevion Medical Systems as the director of project management and helped the company secure its initial FDA 510(k) clearance. In 2012, he joined Sun Nuclear Corporation as the product manager of its Patient QA products. He continues to work for Sun Nuclear today as the senior director of research and regulatory.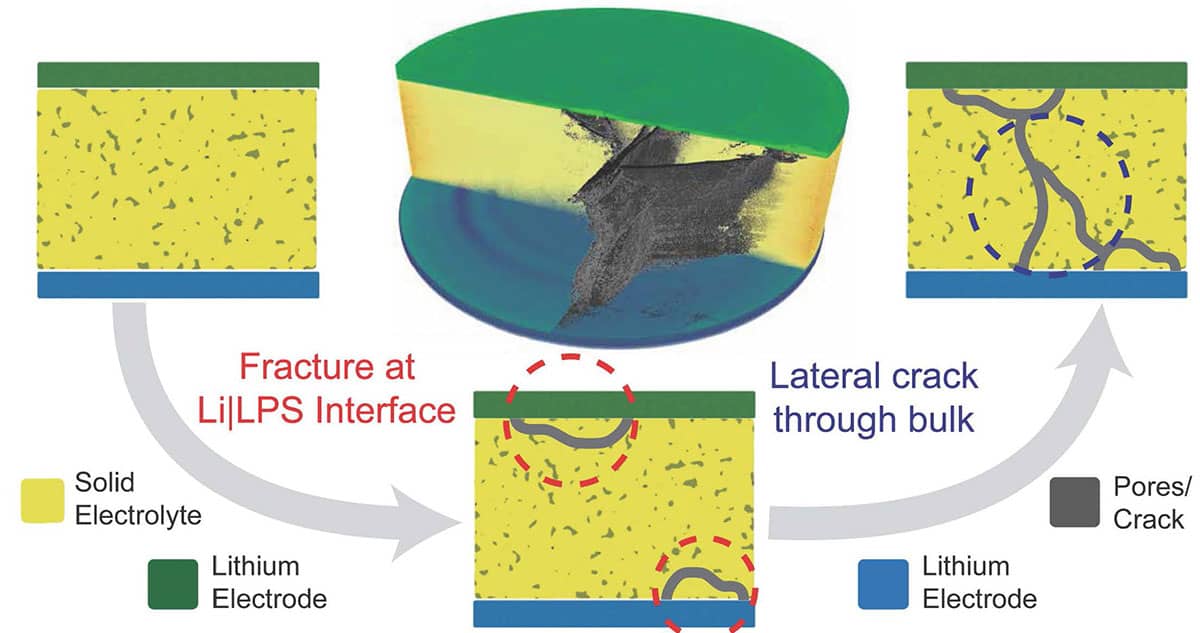 Transportation accounts for 23% of energy-related carbon dioxide emissions and electrification is a pathway toward ameliorating these growing challenges. All solid-state batteries could potentially address the safety and driving range requirements necessary for widespread adoption of electric vehicles. However, the power densities of all solid-state batteries are limited because of ineffective ion transport at solid–solid interfaces.
Transportation accounts for 23% of energy-related carbon dioxide emissions and electrification is a pathway toward ameliorating these growing challenges. All solid-state batteries could potentially address the safety and driving range requirements necessary for widespread adoption of electric vehicles. However, the power densities of all solid-state batteries are limited because of ineffective ion transport at solid–solid interfaces. Kelsey Hatzell is assistant professor of mechanical engineering, assistant professor of chemical and biomolecular engineering, and Flowers Family Dean’s Faculty fellow in engineering at Vanderbilt University. Her research focuses on printable materials and understanding electrochemistry at interfaces as well as solution-processed material synthesis of low-dimensional materials for energy storage and water desalination application. She earned her BS/BA in engineering/economics from Swarthmore College; MS in mechanical engineering from Pennsylvania State University, US; and PhD in material science and engineering at Drexel University, US. Hatzell has received research awards including the Sloan Fellowship in Chemistry (2020), POLiS Award for Excellence (2021), MRS Nelson “Buck” Robinson Science and Technology Award for Renewable Energy (2019), ECS Toyota Young Investigator Fellowship (2019), Oak Ridge Associated Universities (ORAU) Ralph E Powe Award (2017), and NSF CAREER Award (2018). She was also a Scialog research fellow in energy storage and negative emissions technologies. She joins the faculty of Princeton University School of Engineering and Applied Science and the Andlinger Center for Energy and the Environment (ACEE) in the fall.
Kelsey Hatzell is assistant professor of mechanical engineering, assistant professor of chemical and biomolecular engineering, and Flowers Family Dean’s Faculty fellow in engineering at Vanderbilt University. Her research focuses on printable materials and understanding electrochemistry at interfaces as well as solution-processed material synthesis of low-dimensional materials for energy storage and water desalination application. She earned her BS/BA in engineering/economics from Swarthmore College; MS in mechanical engineering from Pennsylvania State University, US; and PhD in material science and engineering at Drexel University, US. Hatzell has received research awards including the Sloan Fellowship in Chemistry (2020), POLiS Award for Excellence (2021), MRS Nelson “Buck” Robinson Science and Technology Award for Renewable Energy (2019), ECS Toyota Young Investigator Fellowship (2019), Oak Ridge Associated Universities (ORAU) Ralph E Powe Award (2017), and NSF CAREER Award (2018). She was also a Scialog research fellow in energy storage and negative emissions technologies. She joins the faculty of Princeton University School of Engineering and Applied Science and the Andlinger Center for Energy and the Environment (ACEE) in the fall.
