Alpha particles are a powerful cancer-killing tool, directly damaging tumour cell DNA regardless of the level of oxygenation or cell cycle stage. The downside of alpha particles is their extremely short range (40-90 µm) in tissue. Now, Israeli start-up company Alpha Tau Medical has created a method to overcome this range limit and showcased its technology at the recent ESTRO 37 congress in Barcelona.
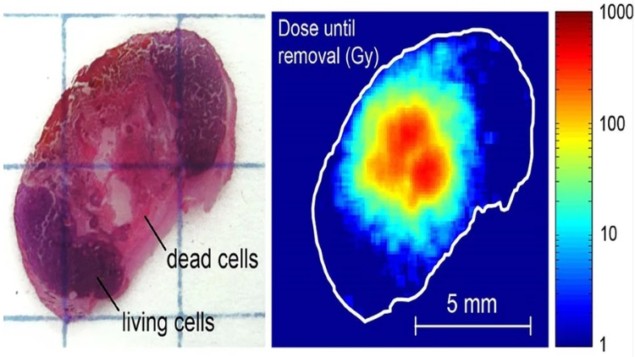
The company’s Alpha DaRT (diffusing alpha-emitters radiation therapy), invented by Itzhak Kelson and Yona Keisari from Tel Aviv University, is based around a radioactive seed containing 224Ra atoms. The seed is injected into a solid tumour and as it decays, it continually releases short-lived daughter atoms (220Rn, 216Po, 212Bi and 212Po), which are also alpha emitters. These atoms diffuse into the tumour, where they emit high-energy alpha particles that destroy tumour tissue. This approach increases the treatment range to a radius of several millimetres.
“Instead of directly irradiating the tumour, 224Ra decays and pushes daughters into the tumour. These are alpha emitters, which start to diffuse and decay again,” explained Amnon Gat, chief operating officer at Alpha Tau. “This enables clinical use of alpha for tumour destruction.”
The 224Ra atoms are fixed onto the seed, so they don’t diffuse into tissue themselves. The daughter atoms diffuse well in the tumour but hardly in healthy tissue, making the treatment highly conformal with no systemic side effects. Alpha radiation also has a high relative biological effectiveness, so less dose is required to induce damage. Another advantage is that because alpha particles are not impacted by oxygen level, Alpha DaRT can treat hypoxic tumours that are resistant to other type of radiation.
Into the clinic
Preclinical trials demonstrated that Alpha DaRT is effective and safe for treating a range of solid tumours, including squamous cell, colon, prostate, brain, pancreatic and lung carcinomas. The therapy is now undergoing clinical trials in Israel and Italy, and has been used to treat 16 patients to date.

The initial trial is examining superficial indications – squamous cell carcinoma of the skin and oral cavity. In such cases, the seeds are placed temporarily (under local anaesthesia) into the lesion using minimally invasive applicators and are removed after 15 days. Thanks to the short half-life of 224Ra (3.7 days), Alpha DaRT can be applied as a single-session treatment that achieves clinical outcome within a few days.
Early results from this feasibility study have demonstrated complete local control of about 80% and zero systemic toxicity. Gat notes that the company also has protocols in place for treatment of pancreatic and prostate cancers. For such deep-seated tumours, the biocompatible seeds are simply left in place.
“Alpha Tau Medical is now focused on starting clinical trials of 12 different protocols at 55 centres in 24 countries around the world,” stated Gat. “The company also plans to open production facilities in each of the key markets, to ensure the supply of the Alpha DaRT.”



