A first-in-human study from researchers at the Mayo Clinic has shown how proton therapy could provide a new treatment option for patients with hard-to-treat ventricular tachycardia (VT), a life-threatening heart rhythm disorder. In the small group of patients examined in this early feasibility study, the treatment led to a 79% reduction in VT episodes.
VT is a type of abnormal heartbeat in which faulty electrical signals in the ventricles cause the heart to beat too quickly, meaning that it can’t pump enough blood around the body. Treatments include antiarrhythmic drugs or the use of catheter ablation to destroy the areas of myocardium (cardiac muscle) responsible for the abnormal signals. Sufferers can also be fitted with an implantable cardioverter-defibrillator (ICD) that automatically delivers a shock to reset the heart’s rhythm during a VT attack.
Some patients, however, don’t respond to conventional therapies, including antiarrhythmic medications and catheter ablations, and ICD shocks can significantly impact quality-of-life. For these cases, cardiac radioablation – which uses external-beam radiotherapy to target the problematic myocardium – is under investigation as an alternative, catheter-free treatment for VT.
Previous clinical studies of cardiac radioablation have employed photon-based irradiation, which can expose surrounding cardiac tissue to low-to-moderate radiation doses. Beams of protons, on the other hand, deposit almost all dose at a defined depth (the Bragg peak) and could enable more precise targeting with reduced irradiation of nearby healthy tissue.
“The main motivation for investigating cardiac radioablation is to improve upon the limitations and suboptimal outcomes of catheter ablation of VT in some patients,” explains lead investigator Konstantinos Siontis. “The motivation specific to protons is the potential dosimetric advantage, allowing more precise myocardial targeting while minimizing radiation to surrounding cardiac and extracardiac structures compared with photons.”
In this new study, reported in Heart Rhythm, Siontis and colleagues used proton-based cardiac radioablation to treat seven patients with advanced cardiomyopathy (disease of the heart muscle) and recurrent VT despite drug treatment and previous catheter ablations.
First-in-human investigation
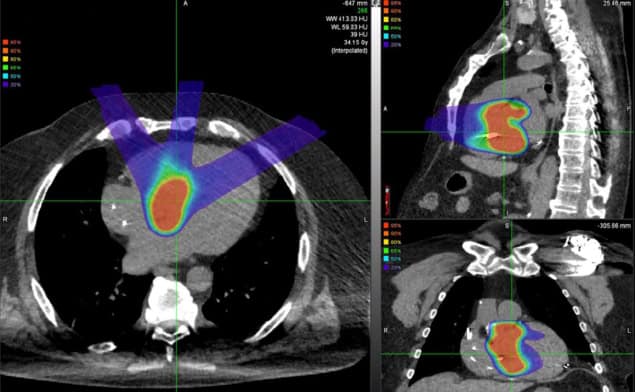
To define the target myocardium for radioablation, the team integrated data from multiple imaging modalities (primarily MRI, plus CT) with information from electrocardiogram (ECG) and electrophysiology mapping originating from the patient’s prior invasive ablation procedures. The CT images were then used to contour the target and organs-at-risk (OARs) and for treatment planning.
The researchers designed treatment plans to deliver a single 30 Gy fraction of expiration-gated intensity-modulated proton therapy to the cardiac internal target volume (ITV, the target myocardium expanded to include cardiac motion) while sparing surrounding OARs. They point out that, due to safety uncertainties in thisfirst-in-human study, they took a generally conservative approach to target definition. In all patients, at least 90% of the ITV received 100% of the prescription dose, while a median of 96.2% of the ITV received at least 95%. Importantly, only 4.3% of non-target myocardium received a dose of 20 Gy or above.
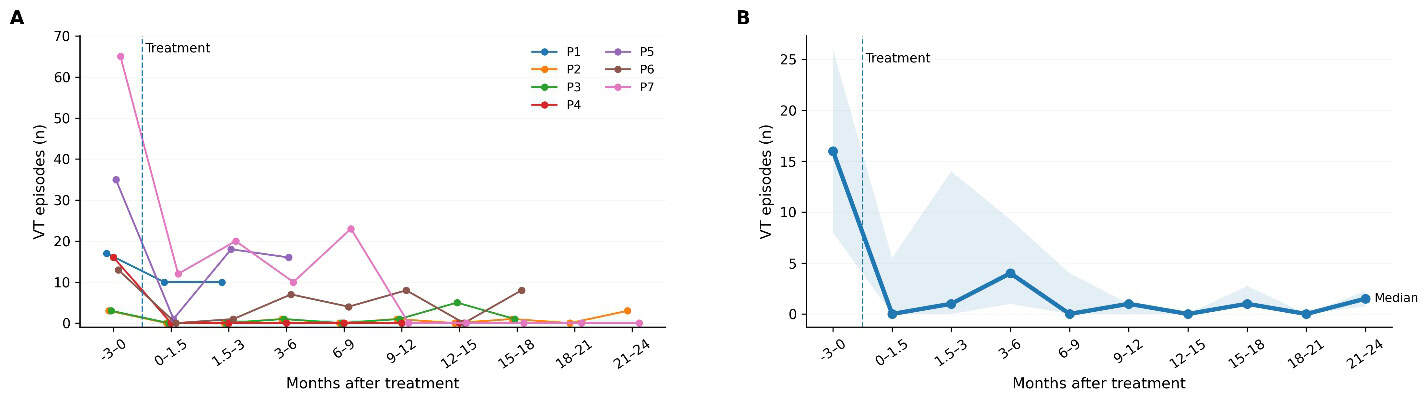
After treatment, the investigators performed follow-up evaluations for up to two years (median 514 days). Most patients experienced recurrent VT during this time, although less frequently than before the radioablation. Across all patients, the rate of VT events declined from 7.24 per patient-month in the three months before treatment to 1.52 per patient-month afterwards – corresponding to a 79% reduction in VT event rate. None of the group experienced serious treatment-related side effects and key heart function measures remained largely stable.
All patients in this study had advanced structural heart disease with severely reduced ventricular function and recurrent VT, putting them at high risk of both arrhythmic and heart failure-related mortality. In line with this profile, two patients required heart transplantation (at 66 and 514 days after treatment) and three died (at 155, 502 and 529 days), due to progressive heart failure.
“This early feasibility study demonstrates that proton cardiac radioablation for refractory VT can be safely planned and delivered with encouraging reductions in arrhythmic burden and no clear treatment-related toxicity,” the researchers conclude. “These findings support the feasibility of proton-based cardiac radioablation and justify further investigation,” they write.
Radiation can reverse heart rhythm disorders by reprogramming damaged cardiac cells
Siontis notes that alongside the emergence of cardiac radioablation techniques, catheter ablation tools are also constantly improving. “Radioablation is unlikely to replace catheter ablation broadly, but it could become an important complementary or salvage option for patients with refractory VT who are poor candidates for invasive procedures,” he tells Physics World.
The team is now planning a larger prospective trial to better define the safety, efficacy and optimal targeting. “We are also investigating improved radiation delivery techniques, such as optimizing treatment planning around cardiac motion,” says Siontis. “In parallel, we continue to investigate photon radioablation in a pivotal randomized trial (RADIATE-VT), while we also offer proton therapy as a compassionate use option for patients in need in our clinical practice.”




