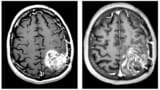
A non-invasive cancer therapy known as tumour treating fields (TTFields) uses low-intensity alternating electric fields to inhibit cancer cell division and cause cell death. A new study providing fresh insights into how the applied electric fields kill cancer cells could help optimize future treatment of the brain cancer glioblastoma (GBM).
Most patients with GBM will have surgery to remove as much of their tumour as possible, before undergoing radiotherapy and chemotherapy. For newly diagnosed or recurrent GBM, tumour growth or spread can sometimes be slowed by adding in TTFields treatment. TTFields directs low-intensity (1–3 V/cm) alternating electric fields through the scalp to the tumour via insulated ceramic transducer arrays. The 200–300 kHz frequencies precisely target the rapidly dividing GBM cells, creating biophysical forces that disrupt cell division.
Simultaneously, the interaction of the electric fields with local conductive biological tissue heats those areas to between 38 and 39.5°C. While careful thermal management is required to prevent this “intrinsic mild hyperthermia (iMH)” side effect from injuring the scalp, studies in pancreatic cancer models have shown that deliberate application of additional hyperthermia in combination with TTFields can enhance cytotoxicity and inhibit cell migration. In this latest study, a team of researchers, led by Aili Zhang from Shanghai Jiaotong University in China, investigated whether the intrinsic heating during TTFields treatments for GBM could be optimized to produce similar advantageous effects.
“Our initial interest was to understand the biological effect of the electric field itself to find out why TTFields therapy works for some people but not for others,” explains Zhang. “As we looked more closely, we found that applying the field inevitably generates heat, which makes it difficult to distinguish the pure electrical effect from the accompanying thermal effect.”
That problem led the team to focus on electrothermal decoupling, in other words, separating the electrical and thermal components: an important step for clarifying the exact mechanism by which the GBM cancer cells are killed and for developing possible treatment optimization protocols.
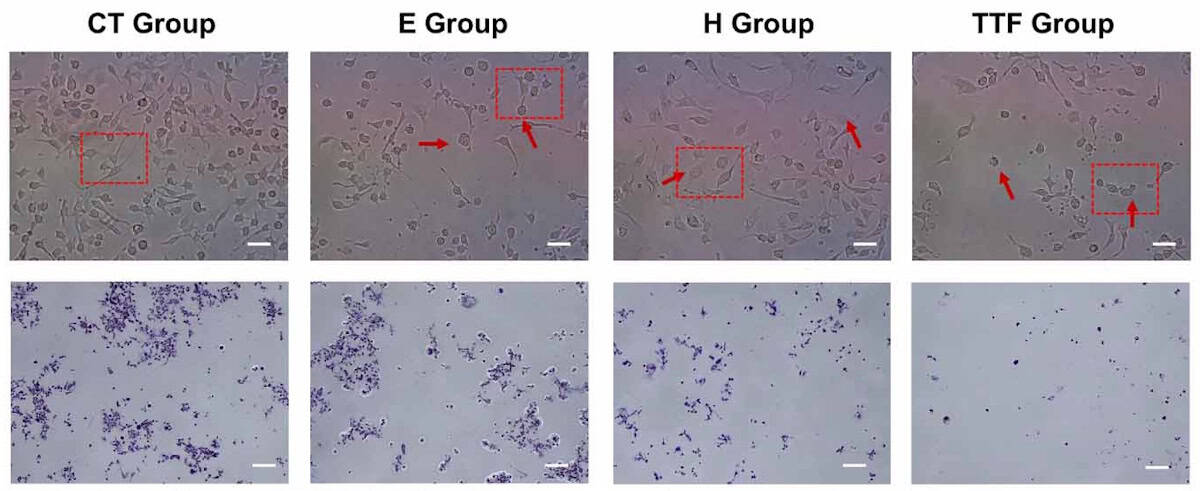
As detailed in Physics in Medicine & Biology, Zhang and colleagues used numerical simulations to help them create an in vitro experimental platform capable of decoupling TTFields’ electrical and thermal components. The 230 kHz, 2 V/cm electric fields were applied via custom-designed, conductive, 2 mm-wide titanium electrodes created to safely enable precise delivery to in vitro wells containing murine GBM cells.
The team used numerical modelling to estimate temperature, and to optimize the electrode geometry and spacing such that a stable and sufficiently uniform electric field could be generated in the central monitoring area where the cells were being analysed.
“Just as importantly, the modelling told us how much intrinsic heating would be generated during TTFields exposure and how to compensate for it. Based on those results, we could set the incubator conditions to create a pure electric condition, a pure thermal condition and the combined TTFields condition,” explains Zhang.
Their results revealed that while the electric field component of TTFields was more closely associated with suppressing the proliferation and migration of cells, the decrease in both cell viability – thanks to elevated levels of calcium ions which help mediate cell death – and metabolic activity was primarily due to the thermal iMH.
“We became genuinely excited when the decoupled experiments started to show that the electric and thermal components were not simply producing the same biological effect at different intensities, but were contributing in clearly different ways,” Zhang tells Physics World.
Tumour-specific radiofrequency fields suppress brain cancer growth
“That was a significant moment because it suggested that the heat generated during TTFields should not be viewed only as an unwanted by-product, but as a potentially meaningful therapeutic component,” she continues, explaining that, importantly, “the combined TTFields condition performed better than would be expected from a simple additive effect”. This electrothermal synergy, the researchers believe, comes from the thermal component sensitizing the cells by increasing membrane vulnerability and disturbing calcium homeostasis, thereby allowing the electric field to more effectively drive cell death.
Next, Zhang plans to confirm the synergistic effect at the molecular level and “systematically examine how the electrothermal interaction changes across different TTFields frequencies, field strengths and thermal conditions for different GBM human cell lines”. The ultimate aim is to find the “optimized treatment protocol” for glioblastoma. Understanding such heating effects could also help optimize other medical treatments such as cardiac ablation, she adds.