The ability to perform functional MRI (fMRI) during deep brain stimulation (DBS) is important for understanding the effects of DBS therapies. DBS, which involves electrical stimulation of neural tissues via implanted electrodes, is used for treatments of movement disorders such as Parkinson’s disease and essential tremor. But the therapeutic mechanisms and neuromodulatory effects of DBS are not well understood.
While fMRI provides a powerful tool for mapping brain activity, strong magnetic field interference from conventional metal DBS electrodes creates artefacts in the MR images. Such artefacts preclude functional and structural mapping of large volumes of brain tissues surrounding the electrodes and impede visualization of local responses at the stimulation site during simultaneous DBS and fMRI.
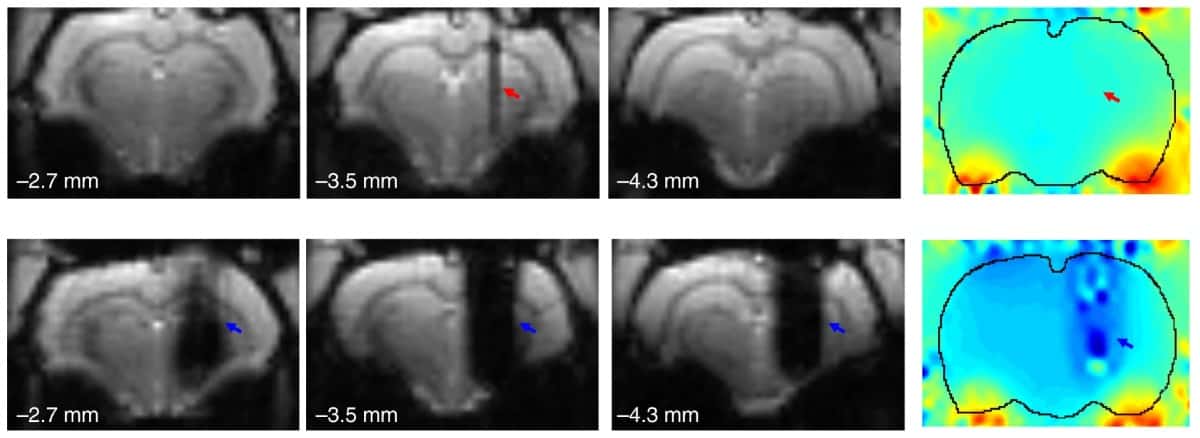
A team in China has now shown that use of novel graphene fibre (GF) DBS microelectrodes can enable full activation pattern mapping during DBS–fMRI in a rat model of Parkinson’s disease. The GF electrodes caused minimal interference with the magnetic field of a 9.4 T MRI scanner, enabling significantly better, artefact-free or near artefact-free, images than those created when using tungsten wire or platinum–iridium electrodes.
The researchers, at Peking University and the Institute of Neuroscience, Chinese Academy of Sciences, fabricated GF electrodes with a charge-injection-capacity 70 times greater than DBS electrodes made of platinum–iridium, the material most commonly used in clinical neural stimulation devices. This high charge-injection-capacity is desirable to decrease MRI artefact size and improve stimulation resolution.

Co-principal investigators Xiaojie Duan and Zhifeng Liang created the GFs from aqueous graphite oxide suspensions injected into a glass pipeline. They baked the pipeline at 230°C to produce a GF matching the pipe geometry, with a fibre diameter of about 75 µm.
To fabricate a GF-stimulating microelectrode, they pasted together two parallel-aligned insulated GFs, with one end soldered to a custom MRI-compatible copper connector that interfaces with the stimulation pulse generator. The GFs were then mechanically cut to expose their cross sections as electrically active sites. These GF electrodes showed a higher charge-injection limit than most available electrode materials.
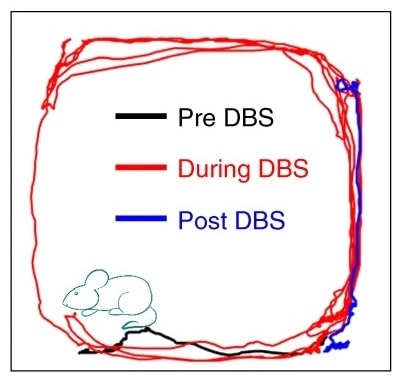
The researchers tested the DBS capability of the GF electrodes by using them to stimulate the subthalamic nucleus in Parkinsonian rats, with stimulation parameters replicating a clinical DBS setting. They observed significant improvement in the rats’ mobility, confirming the therapeutic efficacy of subthalamic nucleus-DBS with GF electrodes.
Comparing MRI artefacts generated by the GF microelectrodes with those created by platinum-iridium microelectrodes of the same diameter showed that the GF electrodes exhibited much smaller artefacts in both anatomical images and functional (echo planar imaging) scans. The GF electrodes also showed high stability under continuous overcurrent pulsing.
“The GF electrodes caused minimal interference to the magnetic field, and their presence would not cause significant attenuation in fMRI signals, thus enabling a full and unbiased mapping of the activation pattern under DBS–fMRI studies,” write the researchers. “Such advantage is critical for exploring the neuromodulatory effects and mechanisms of DBS therapies.”
“With the unique capability for full and unbiased mapping of the entire circuit and network connectivity without obstructing brain nuclei, future DBS–studies with the GF electrodes at different targets and with varied stimulation frequency and strength could provide important insights into brain circuitries and network connections, as well as the therapeutic mechanisms underlying various DBS therapies,” they write.
The team now hopes to use the GF electrodes for DBS–fMRI studies on other neurological disorders, such as treatment-resistant depression.
The study details are reported in Nature Communications.



